OPES walk-in QA, RA, F&D, ADL

- Company Overview
- Job Role & Responsibilities
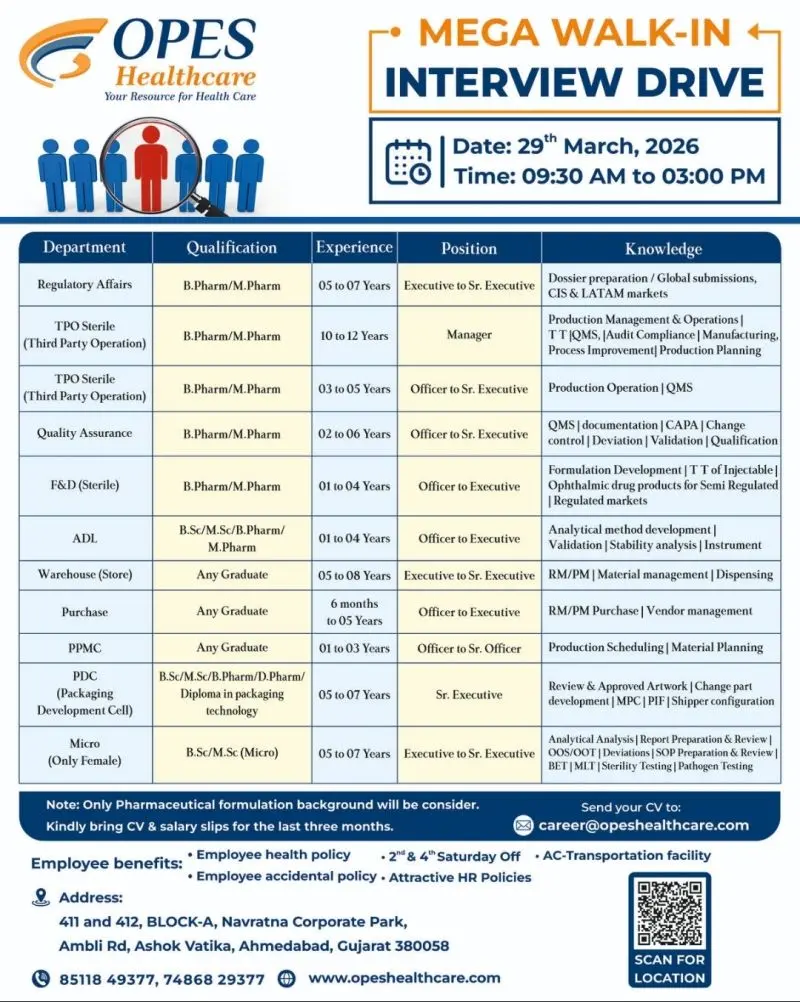
- Regulatory Affairs (Executive to Sr. Executive)
- TPO Sterile (Manager / Officer to Sr. Executive)
- Quality Assurance (Officer to Sr. Executive)
- F&D – Sterile (Officer to Executive)
- ADL – Analytical Development Lab
- Warehouse (Store)
- Purchase
- PPMC (Production Planning & Material Control)
- PDC (Packaging Development Cell)
- Microbiology (Only Female)
- Eligibility / Qualifications
- Required Education
- Experience Requirements
- Location & Salary
- Location:
- Salary Insights:
- Employee Benefits:
- Walk-In Interview Details
- Application Process
- Documents Required:
- Why This Opportunity Stands Out
- SEO Keywords Integrated Naturally
- FAQs
- Is this a walk-in interview?
- Who can apply for regulatory affairs roles?
- Are freshers eligible?
- What is the scope of regulatory affairs careers?
- What documents should I carry?
- Summary Table
BPharm MPharm 25+ Pharma Jobs Ahmedabad Walk-In
BPharm/MPharm hiring for 25+ roles at OPES Healthcare Ahmedabad. QA, RA, F&D, ADL. Walk-in 29 March 2026.
OPES Healthcare is conducting a mega walk-in interview drive in Ahmedabad for multiple departments including Regulatory Affairs, Quality Assurance, Formulation & Development (F&D), ADL, Warehouse, Purchase, and Production Planning. This is a strong opportunity for candidates aiming for pharmaceutical formulation jobs, regulatory affairs careers, and quality assurance roles in a fast-growing pharma environment.
If you are searching for pharma jobs in Ahmedabad, regulatory affairs jobs India, or high-paying pharmaceutical quality assurance careers, this hiring drive is highly relevant. The company is hiring across mid-level and senior roles, making it suitable for experienced professionals looking for career advancement.
These roles fall under high CPC domains such as regulatory affairs jobs, pharma QA jobs, formulation development careers, and pharmaceutical quality systems, all of which offer strong salary potential and long-term career growth.
Company Overview
OPES Healthcare is a pharmaceutical formulation-focused company specializing in regulated and semi-regulated markets. The company has built a reputation for quality manufacturing, regulatory compliance, and global submissions across markets such as CIS and LATAM.
The organization operates with strong quality systems (QMS), advanced formulation capabilities, and a structured approach to product development and commercialization. Their work spans injectable and ophthalmic dosage forms, which require high precision and regulatory adherence.
Working at OPES Healthcare provides exposure to:
- Regulatory submissions and dossier preparation
- Sterile manufacturing and formulation development
- Quality assurance systems (QMS, CAPA, deviation control)
- Analytical method development and validation
These are high-value skill areas aligned with global pharma career opportunities and regulatory-driven roles.
Job Role & Responsibilities
OPES Healthcare is hiring across multiple core pharmaceutical departments. Each role contributes directly to drug quality, regulatory approval, and manufacturing efficiency.
Regulatory Affairs (Executive to Sr. Executive)
- Prepare and review dossiers for global submissions
- Handle regulatory documentation for CIS & LATAM markets
- Ensure compliance with international regulatory guidelines
TPO Sterile (Manager / Officer to Sr. Executive)
- Manage third-party manufacturing operations
- Oversee production planning and execution
- Ensure compliance with quality and audit standards
- Drive process improvements and operational efficiency
Quality Assurance (Officer to Sr. Executive)
- Handle QMS documentation and compliance
- Manage CAPA, deviation, and change control processes
- Support validation and qualification activities
F&D – Sterile (Officer to Executive)
- Work on formulation development of injectable and ophthalmic products
- Support technology transfer (TT) processes
- Develop products for regulated and semi-regulated markets
ADL – Analytical Development Lab
- Perform analytical method development and validation
- Conduct stability studies and analytical testing
- Handle instruments and documentation
Warehouse (Store)
- Manage raw material and packaging material (RM/PM)
- Handle dispensing and inventory control
Purchase
- Manage procurement of RM/PM
- Handle vendor management and negotiations
PPMC (Production Planning & Material Control)
- Plan production schedules
- Manage material requirements and coordination
PDC (Packaging Development Cell)
- Review and approve packaging artwork
- Handle packaging development and configuration
Microbiology (Only Female)
- Perform sterility testing and microbial analysis
- Handle OOS/OOT investigations and documentation
- Conduct pathogen testing and report preparation
These roles are essential for pharmaceutical product lifecycle management, from development to commercialization.
Eligibility / Qualifications
Required Education
Candidates must have a pharmaceutical or relevant academic background.
Accepted courses include:
B.Pharm, M.Pharm, BSc, MSc, Microbiology, Biotechnology, Biochemistry, Life Sciences, D.Pharm, Diploma in Packaging Technology, Any Graduate (for support roles)
Experience Requirements
- Regulatory Affairs: 5–7 years
- TPO Sterile Manager: 10–12 years
- TPO Sterile Executive: 3–5 years
- QA: 2–6 years
- F&D: 1–4 years
- ADL: 1–4 years
- Warehouse: 5–8 years
- Purchase: 6 months–5 years
- PPMC: 1–3 years
- PDC: 5–7 years
- Microbiology: 5–7 years
Note: Only candidates with pharmaceutical formulation background will be considered.
Location & Salary
Location:
- Ahmedabad, Gujarat
Salary Insights:
Salary is not disclosed, but these roles fall under high-paying pharma domains such as:
- Regulatory affairs salary India
- QA executive pharma salary
- Formulation development scientist salary
- Pharmaceutical industry jobs salary growth
Experienced candidates in regulatory and sterile domains typically receive premium packages.
Employee Benefits:
- Attractive HR policies
- Employee health and accidental insurance
- 2nd and 4th Saturday off
- Transportation facility
Walk-In Interview Details
- Date: 29th March 2026
- Time: 09:30 AM to 03:00 PM
- Venue: 411 & 412, Block-A, Navratna Corporate Park, Ambli Road, Ashok Vatika, Ahmedabad – 380058
Application Process
Candidates can directly attend the walk-in interview.
Alternatively, send your CV to:
Email: career@opeshealthcare.com
Contact: 85118 49377 / 74868 29377
Documents Required:
- Updated CV
- Last three months salary slips
- Educational and experience documents
Why This Opportunity Stands Out
- Multiple openings across core pharma departments
- Strong focus on regulatory and sterile formulation roles
- Exposure to global markets (CIS & LATAM)
- Growth opportunities in QA, RA, and F&D
- Work in a compliance-driven pharma environment
These roles are ideal for professionals aiming for careers in pharmaceutical development, regulatory affairs, and quality systems.
SEO Keywords Integrated Naturally
pharma jobs Ahmedabad, regulatory affairs jobs India, QA pharma jobs, formulation development jobs, sterile pharma jobs, pharmaceutical industry careers, high paying pharma jobs India, biotech jobs Ahmedabad
FAQs
Is this a walk-in interview?
Yes, this is a direct walk-in hiring drive in Ahmedabad.
Who can apply for regulatory affairs roles?
Candidates with B.Pharm or M.Pharm and relevant experience in dossier preparation and submissions.
Are freshers eligible?
Most roles require experience, but some entry-level roles like purchase may accept candidates with minimal experience.
What is the scope of regulatory affairs careers?
Regulatory affairs is one of the highest-paying pharma career paths with global opportunities.
What documents should I carry?
Updated CV, salary slips, and all academic and experience certificates.
Summary Table
| Company | OPES Healthcare |
|---|---|
| Vacancies | Regulatory Affairs Executive/Sr Executive, TPO Sterile Manager/Executive, QA Officer/Sr Executive, F&D Executive, ADL Officer, Warehouse Executive, Purchase Officer, PPMC Officer, PDC Sr Executive, Microbiology Executive |
| Required Education | B.Pharm, M.Pharm, BSc, MSc, Microbiology, Biotechnology, Biochemistry, Life Sciences, D.Pharm, Diploma Packaging Technology, Any Graduate |
| Experience | 6 months to 12+ years depending on role |
You must sign in to apply for this position.


