Apnar Walk-In Production, QA, QC & Microbiology

- Company Overview
- Job Role & Responsibilities
- Production Department
- Packaging Department
- Quality Control (QC)
- Microbiology
- Quality Assurance (QA)
- Validation (Central Lab)
- Eligibility / Qualifications
- Required Education
- Relevant Courses
- Experience
- Skills Required
- Location & Salary
- Job Location
- Salary
- Application Process
- Walk-In Interview Details
- Alternative Application
- Contact Details
- Why This Opportunity Matters
- FAQs
- How many vacancies are available?
- What qualifications are required?
- What experience is required?
- Is USFDA experience required?
- How can I apply?
- Summary Table
Apnar Pharma Walk-In Interview 2026 | 25+ Vacancies in Production, QA, QC & Microbiology
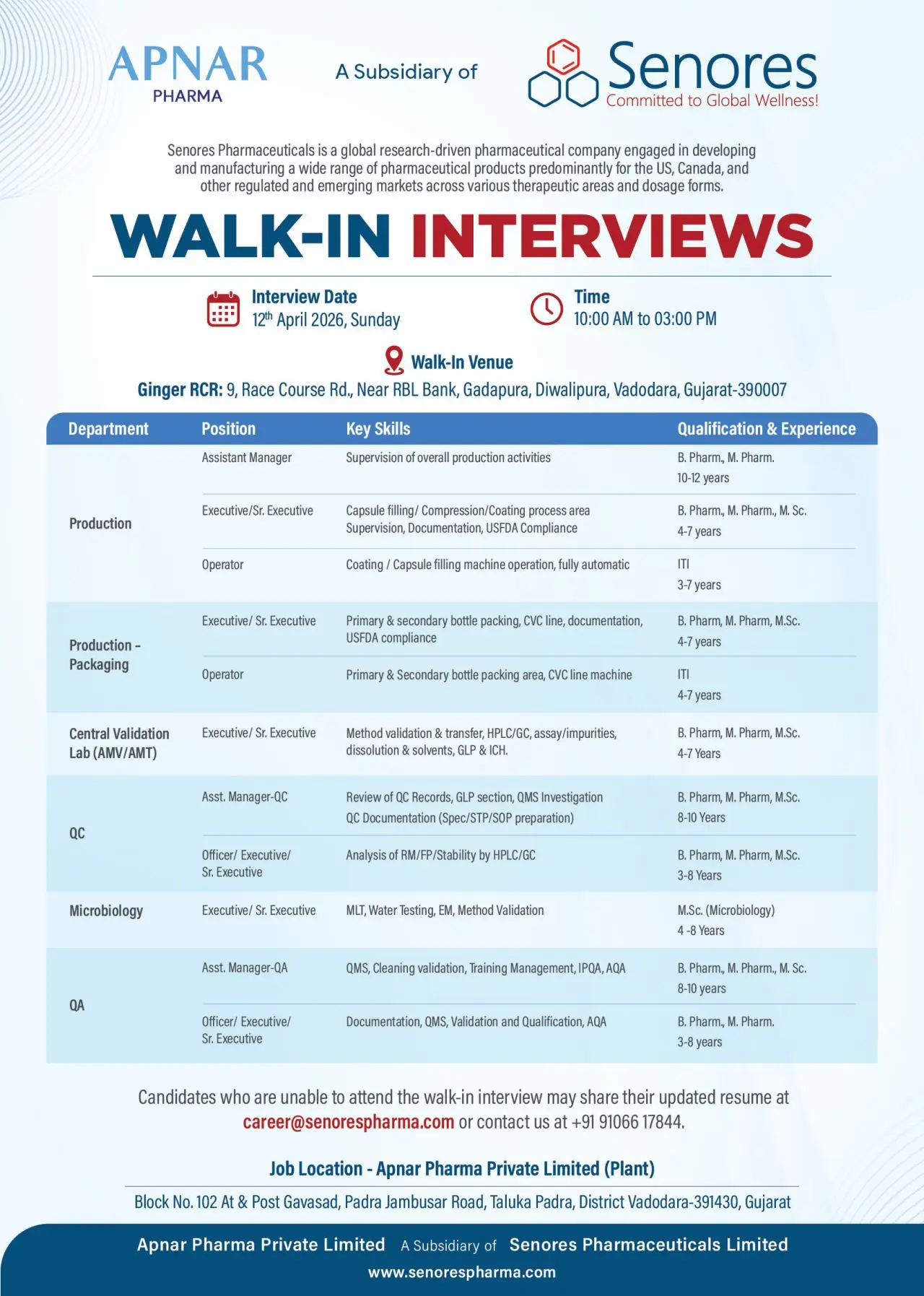
Apnar Pharma Private Limited, a subsidiary of Senores Pharmaceuticals, is conducting a walk-in interview for multiple vacancies across Production, Quality Assurance, Quality Control, Microbiology, and Validation departments at its Vadodara facility. The company is offering more than 25 openings ranging from Operator to Assistant Manager positions, making this a high-value opportunity for experienced pharma professionals.
This hiring drive is scheduled on 12th April 2026 in Vadodara, Gujarat, and is ideal for candidates with 3 to 12 years of experience in regulated pharmaceutical manufacturing, especially those with exposure to USFDA compliance, GMP systems, and formulation production.
Company Overview
Apnar Pharma Private Limited operates under Senores Pharmaceuticals, a globally recognized research-driven pharmaceutical company. The organization focuses on developing and manufacturing high-quality pharmaceutical products for regulated markets such as the US and Canada, along with emerging markets worldwide.
With a strong emphasis on regulatory compliance, innovation, and quality systems, the company provides a robust platform for professionals to grow in areas like formulation development, quality assurance, validation, and pharmaceutical manufacturing operations.
Job Role & Responsibilities
Production Department
- Supervise manufacturing activities including compression, coating, and capsule filling
- Handle documentation and ensure compliance with GMP and USFDA guidelines
- Operate automated equipment and maintain batch production records
Packaging Department
- Manage primary and secondary bottle packing operations
- Handle CVC line operations and packaging documentation
- Ensure regulatory compliance in packaging processes
Quality Control (QC)
- Perform analysis of raw materials, finished products, and stability samples
- Operate analytical instruments such as HPLC and GC
- Maintain documentation, support investigations, and ensure GLP compliance
Microbiology
- Conduct environmental monitoring (EM) and water testing
- Perform microbial limit testing (MLT)
- Support method validation and microbiological analysis
Quality Assurance (QA)
- Handle QMS documentation, validation, and qualification activities
- Manage IPQA, AQA, and cleaning validation processes
- Ensure compliance with regulatory and quality standards
Validation (Central Lab)
- Perform method validation and transfer activities
- Work on analytical validation using HPLC/GC techniques
- Ensure compliance with ICH and GLP guidelines
Eligibility / Qualifications
Required Education
B.Pharm, M.Pharm, M.Sc, ITI
Relevant Courses
Pharmaceutical Sciences, Microbiology, Biotechnology, Analytical Chemistry, Industrial Pharmacy, Quality Assurance, Process Engineering
Experience
- Operator Roles: 3–7 years
- Executive Roles: 3–8 years
- Assistant Manager Roles: 8–12 years
- Experience in USFDA-regulated pharma environment preferred
Skills Required
- Strong knowledge of GMP, GLP, and regulatory compliance
- Experience in pharmaceutical manufacturing and quality systems
- Hands-on expertise in HPLC, GC, validation, or production equipment
- Good documentation and audit readiness skills
Location & Salary
Job Location
Apnar Pharma Pvt. Ltd., Vadodara, Gujarat
Salary
- Competitive salary based on experience and designation
- Additional benefits as per company policy
Application Process
Walk-In Interview Details
- Date: 12th April 2026 (Sunday)
- Time: 10:00 AM to 03:00 PM
- Venue: Ginger Hotel, Race Course Road, Vadodara, Gujarat
Alternative Application
Candidates unable to attend can email their CV to:
Contact Details
- Phone: +91 91066 17844
Why This Opportunity Matters
- Work with a global pharmaceutical company serving regulated markets
- Exposure to USFDA compliance and advanced manufacturing systems
- Career growth in QA, QC, production, and validation domains
- Opportunity to work in high-demand pharma roles with long-term stability
FAQs
How many vacancies are available?
There are 25+ vacancies across multiple departments.
What qualifications are required?
B.Pharm, M.Pharm, M.Sc, and ITI candidates are eligible.
What experience is required?
3 to 12 years depending on the role.
Is USFDA experience required?
Preferred but not mandatory for all roles.
How can I apply?
Attend the walk-in interview or email your CV to the provided address.
Summary Table
| Company | Apnar Pharma Pvt. Ltd. |
|---|---|
| Vacancies | Production, QA, QC, Microbiology, Validation Roles |
| Required Education | B.Pharm, M.Pharm, M.Sc, ITI |
| Experience | 3–12 Years |
You must sign in to apply for this position.


