Akums Walk-in Production, QA, QC

- Company Overview
- Job Role & Responsibilities
- Production (DPI / Nasal / Injectable)
- Quality Assurance (QA)
- Quality Control (QC)
- QC Microbiology
- Engineering (Maintenance / HVAC / Utilities)
- Operator Roles (Production & Engineering)
- Eligibility / Qualifications
- Location & Salary
- Application Process
- Why You Should Apply
- FAQs
- Summary Table
B.Pharm M.Pharm Jobs Akums Haridwar Walk-in
Akums hiring for Production, QA, QC roles in Haridwar. B.Pharm, M.Pharm, MSc candidates with 1–8 yrs experience eligible.
Akums Drugs & Pharmaceuticals Ltd. is conducting a large-scale walk-in interview for multiple vacancies across Production, Quality Assurance (QA), Quality Control (QC), Microbiology, and Engineering departments at its Haridwar Plant. This hiring drive is ideal for professionals with 1 to 8 years of experience looking for pharma jobs in Haridwar, regulatory manufacturing roles, and high-growth opportunities in USFDA-approved pharmaceutical plants.
Candidates searching for B.Pharm jobs in pharmaceutical companies, QC HPLC jobs, QA IPQA roles, or pharma engineering jobs with regulatory exposure should not miss this opportunity. Limited walk-in slots are available, so early reporting is strongly recommended.
Company Overview
Akums Drugs & Pharmaceuticals Ltd. is one of India’s largest contract development and manufacturing organizations (CDMO), known for its strong presence in regulated markets such as USFDA, EU, MHRA, and ANVISA. The company specializes in formulation development, manufacturing, and global supply of pharmaceutical products.
With advanced facilities and strict compliance systems, Akums plays a key role in supporting global healthcare needs. Working at Akums provides exposure to international regulatory standards, GMP manufacturing practices, and cutting-edge pharmaceutical technologies.
Job Role & Responsibilities
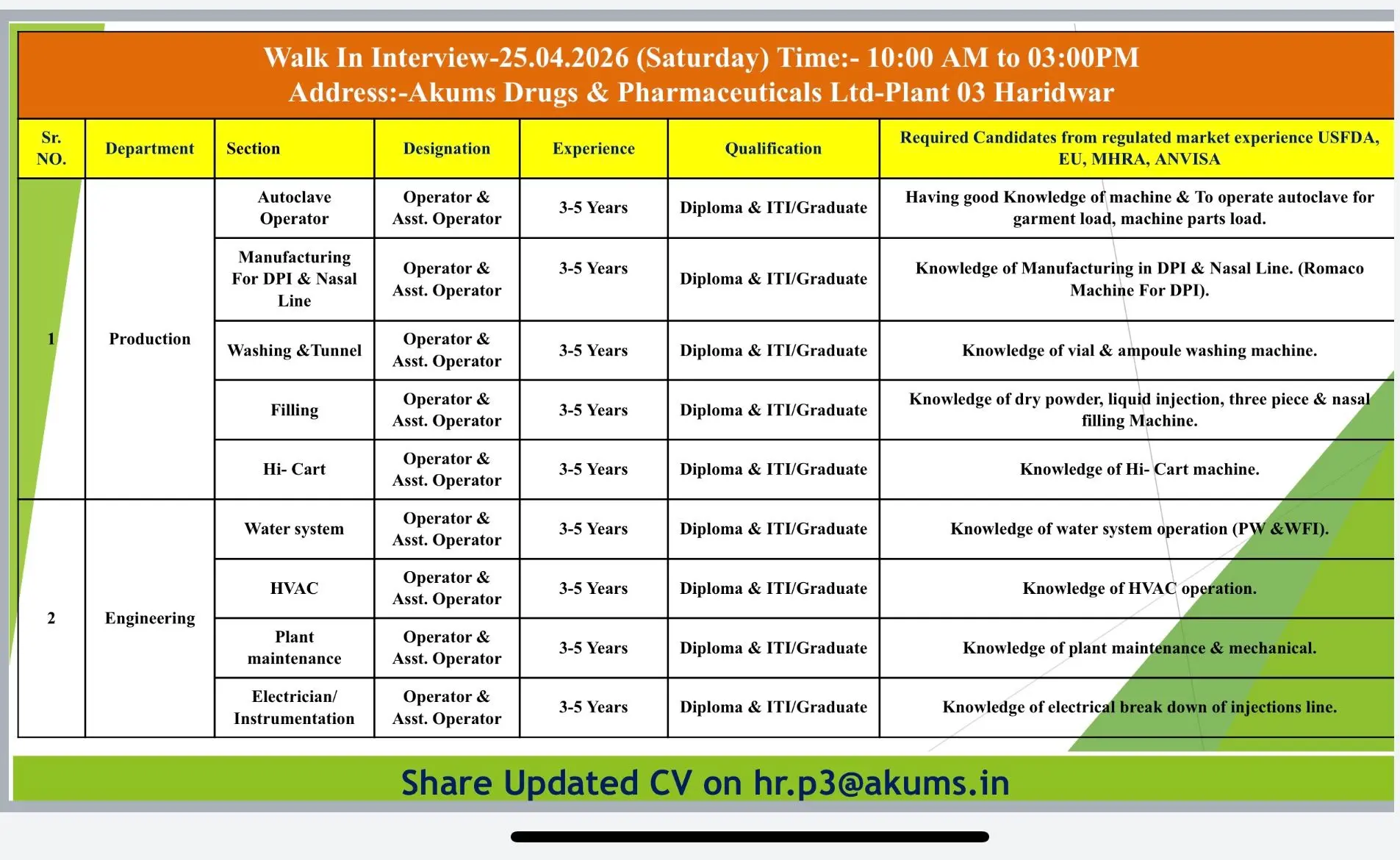
Production (DPI / Nasal / Injectable)
- Handle manufacturing of DPI and nasal formulations using Romaco machines
- Execute production processes as per SOPs and GMP guidelines
- Maintain batch manufacturing records and ensure line clearance
- Operate filling, washing, tunnel, and autoclave systems
- Monitor production parameters and ensure process compliance
Quality Assurance (QA)
- Perform IPQA activities and in-process checks
- Support qualification, validation, and documentation processes
- Ensure compliance with QMS and regulatory standards
- Handle deviations, CAPA, and audit readiness
Quality Control (QC)
- Perform analytical testing using HPLC and other instruments
- Conduct analytical method validation (AMV)
- Ensure compliance with GLP and documentation practices
QC Microbiology
- Conduct water testing, BET, and microbiological analysis
- Maintain laboratory records and compliance systems
Engineering (Maintenance / HVAC / Utilities)
- Handle plant maintenance, HVAC, water systems (PW/WFI)
- Manage breakdown and preventive maintenance
- Ensure smooth operation of utilities and equipment
Operator Roles (Production & Engineering)
- Operate autoclave, filling, washing, and packaging machines
- Handle electrical, instrumentation, and mechanical systems
- Maintain operational logs and ensure safety compliance
These roles require hands-on experience in regulated pharma environments and offer strong exposure to global compliance systems.
Eligibility / Qualifications
Candidates must meet the following qualifications:
M.Pharm, B.Pharm, M.Sc (Chemistry/Microbiology), B.Sc, B.Tech, Diploma, ITI
Relevant courses include: Pharmaceutics, Pharmaceutical Analysis, Microbiology, Mechanical Engineering, Electrical Engineering
- Experience: 1 to 8 years (depending on role)
- Experience in USFDA/EU/MHRA regulated plants preferred
- Strong knowledge of GMP, GLP, and regulatory compliance
- Willingness to work in shift-based operations
Location & Salary
- Job Location: Akums Plant 03, Haridwar
- Experience: 1 to 8 years
- Salary: Competitive and as per industry standards
Application Process
Candidates can attend the walk-in interview directly or apply via email.
Walk-in Details:
Date: 25th April 2026
Time: 10:00 AM to 03:00 PM
Venue:
Akums Drugs & Pharmaceuticals Ltd. – Plant 03
Haridwar
Email Application:
hr.p3@akums.in
Candidates must carry updated resume, educational documents, experience certificates, and salary details.
Apply early or attend the walk-in on time to secure your opportunity.
Why You Should Apply
- Opportunity to work in USFDA-approved pharma company
- Exposure to regulated manufacturing environments
- High-demand roles in QA, QC, and Production
- Strong career growth in pharmaceutical industry
- Hands-on experience with advanced manufacturing technologies
FAQs
Who can apply for this walk-in drive?
Candidates with pharma experience and relevant qualifications.
Is regulatory experience required?
Yes, candidates with USFDA/EU/MHRA experience are preferred.
What departments are hiring?
Production, QA, QC, Microbiology, and Engineering.
Is this a walk-in interview?
Yes, candidates must attend the venue directly.
How to apply?
Attend the walk-in or send your resume via email.
Summary Table
Company Akums Drugs & Pharmaceuticals Ltd.
Vacancies QA Executive, QC Analyst, Production Executive, Engineering Officer
Required Education B.Pharm, M.Pharm, M.Sc, B.Sc, Diploma, ITI
Experience 1 to 8 Years

You must sign in to apply for this position.


