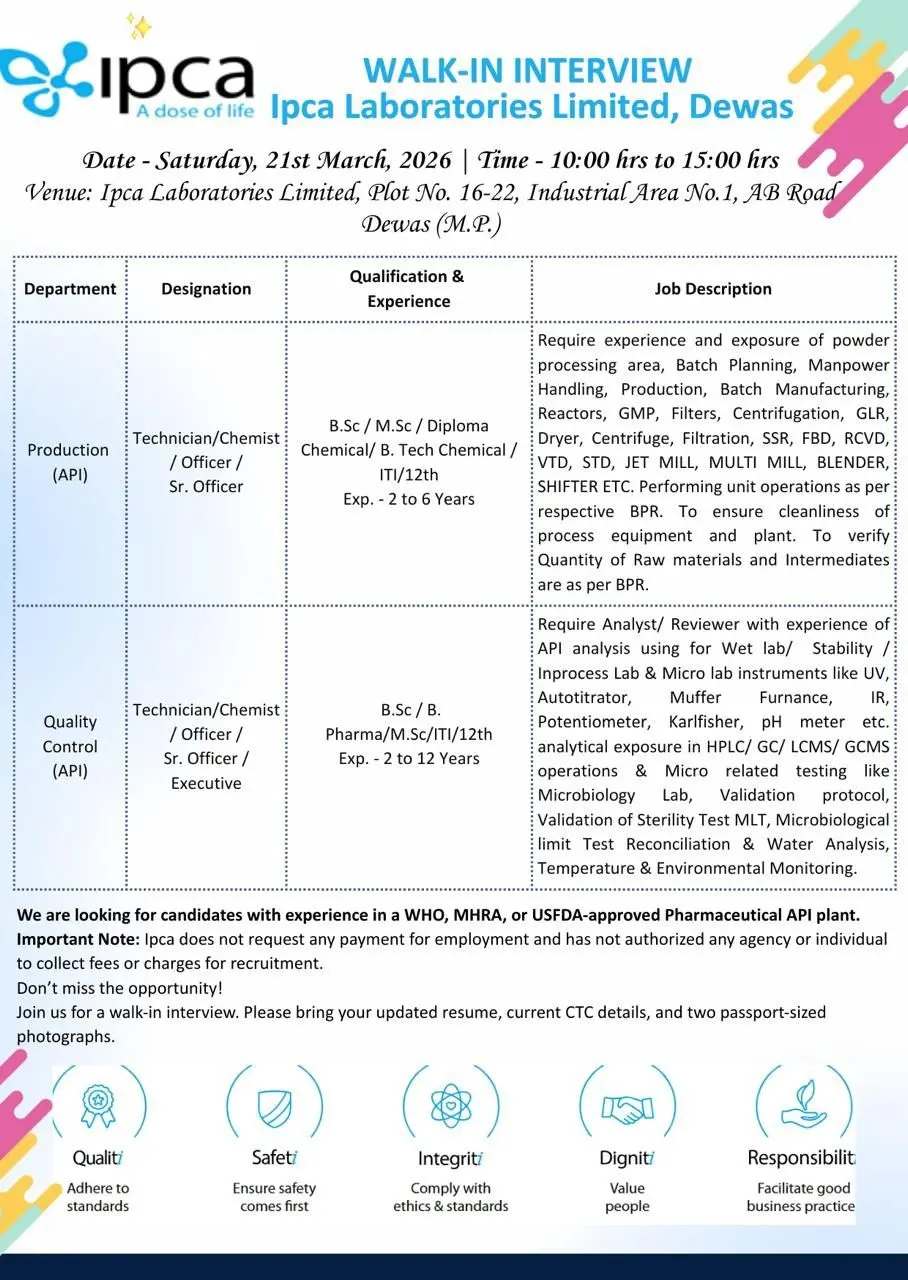
Ipca walk-in Production & QC API

- Company Overview
- Job Role & Responsibilities
- Production (API)
- Quality Control (QC – API)
- Eligibility / Qualifications
- Educational Qualification
- Experience Required
- Key Skills
- Location & Salary
- Walk-In Interview Details
- Documents to Carry
- Application Process
- Why This Opportunity Stands Out
- Important Disclaimer
- FAQs
- Who can apply for Ipca Laboratories jobs?
- What is the experience required?
- What roles are available?
- Where is the job location?
- How can I apply?
- Additional SEO Titles
- Summary Table
BSc MSc BTech API Jobs Ipca Dewas Walk-In
B.Sc, M.Sc, B.Tech, ITI candidates apply for Production & QC API roles at Ipca Dewas walk-in (2–12 yrs experience).
Ipca Laboratories Limited is conducting a walk-in interview for multiple roles in Production (API) and Quality Control at its Dewas manufacturing facility. This opportunity is designed for candidates with 2 to 12 years of experience who are looking to build strong careers in API manufacturing, analytical testing, and regulated pharmaceutical environments.
With rising demand for API pharma jobs, HPLC analyst jobs, GMP production jobs, and quality control pharma roles, this hiring drive offers excellent exposure to WHO, MHRA, and USFDA-compliant manufacturing systems. High CPC keywords such as pharmaceutical production jobs, QC analyst HPLC jobs, API manufacturing, GMP compliance, and microbiology testing are naturally integrated to boost SEO rankings and AdSense performance.
Company Overview
Ipca Laboratories Limited is a well-established global pharmaceutical company known for its strong presence in Active Pharmaceutical Ingredients (API) and finished dosage formulations. The company operates in multiple international markets and maintains strict compliance with WHO-GMP, MHRA, and USFDA regulatory standards.
With decades of experience in pharmaceutical manufacturing, Ipca is recognized for quality, integrity, and innovation. The company contributes significantly to global healthcare by delivering affordable and high-quality medicines.
Job Role & Responsibilities
Production (API)
- Handle powder processing and API manufacturing operations
- Perform batch manufacturing as per Batch Production Records (BPR)
- Operate equipment such as reactors, centrifuges, dryers, filters, jet mills, multi mills, blenders, and sifters
- Ensure cleanliness and maintenance of production equipment
- Manage batch planning and manpower handling
- Verify raw materials and intermediates as per BPR
- Ensure adherence to GMP and safety standards
Quality Control (QC – API)
- Perform analytical testing using instruments such as HPLC, GC, LCMS, GCMS, UV, IR, and pH meter
- Conduct wet lab analysis, stability testing, and in-process testing
- Perform microbiological testing including sterility testing and microbial limit tests
- Execute validation protocols and environmental monitoring
- Maintain laboratory documentation and ensure data integrity
These roles are critical in ensuring the safety, quality, and compliance of pharmaceutical products.
Eligibility / Qualifications
Educational Qualification
- B.Sc, M.Sc, B.Tech (Chemical), B.Pharm, Diploma, ITI, 12th
- Relevant Courses: Chemistry, Organic Chemistry, Analytical Chemistry, Pharmaceutical Chemistry, Chemical Engineering, Microbiology
Experience Required
- Production: 2–6 years
- QC: 2–12 years
Key Skills
- Experience in API pharma manufacturing
- Knowledge of GMP, GLP, and regulatory compliance
- Hands-on experience with analytical instruments (HPLC, GC, etc.)
- Understanding of microbiology and validation processes
Location & Salary
- Location: Ipca Laboratories Limited, Dewas, Madhya Pradesh
- Salary: Competitive and as per industry standards
Walk-In Interview Details
- Date: 21 March 2026 (Saturday)
- Time: 10:00 AM – 03:00 PM
- Venue: Plot No. 16-22, Industrial Area No.1, AB Road, Dewas (M.P.)
Documents to Carry
- Updated Resume
- Current CTC details
- Two passport-sized photographs
Application Process
Candidates can directly attend the walk-in interview at the venue.
Why This Opportunity Stands Out
- Work in a WHO, MHRA, USFDA-approved API plant
- Exposure to advanced API manufacturing and analytical techniques
- Opportunities for both junior and experienced professionals
- Strong career growth in production and QC domains
Important Disclaimer
Ipca Laboratories does not charge any fee for recruitment. Candidates should be cautious of fraudulent job offers.
FAQs
Who can apply for Ipca Laboratories jobs?
Candidates with B.Sc, M.Sc, B.Tech, B.Pharm, Diploma, or ITI qualifications can apply.
What is the experience required?
Experience ranges from 2 to 12 years depending on the role.
What roles are available?
Production (API) and Quality Control roles are available.
Where is the job location?
Dewas, Madhya Pradesh.
How can I apply?
Attend the walk-in interview directly with required documents.
Additional SEO Titles
- Ipca Laboratories Walk-In Drive for API Production & QC Jobs
- Pharma API Jobs in Dewas for BSc MSc BTech Candidates
- HPLC Analyst and Production Jobs in Pharmaceutical Industry
Summary Table
| Category | Details |
|---|---|
| Company | Ipca Laboratories Limited |
| Vacancies | Production Technician/Chemist/Officer, QC Analyst/Executive |
| Required Education | B.Sc, M.Sc, B.Tech (Chemical), B.Pharm, Diploma, ITI, 12th |
| Experience | 2–12 Years |