SynZeal Hiring R&D, RA, IP, QA

- Company Overview
- Job Role & Responsibilities
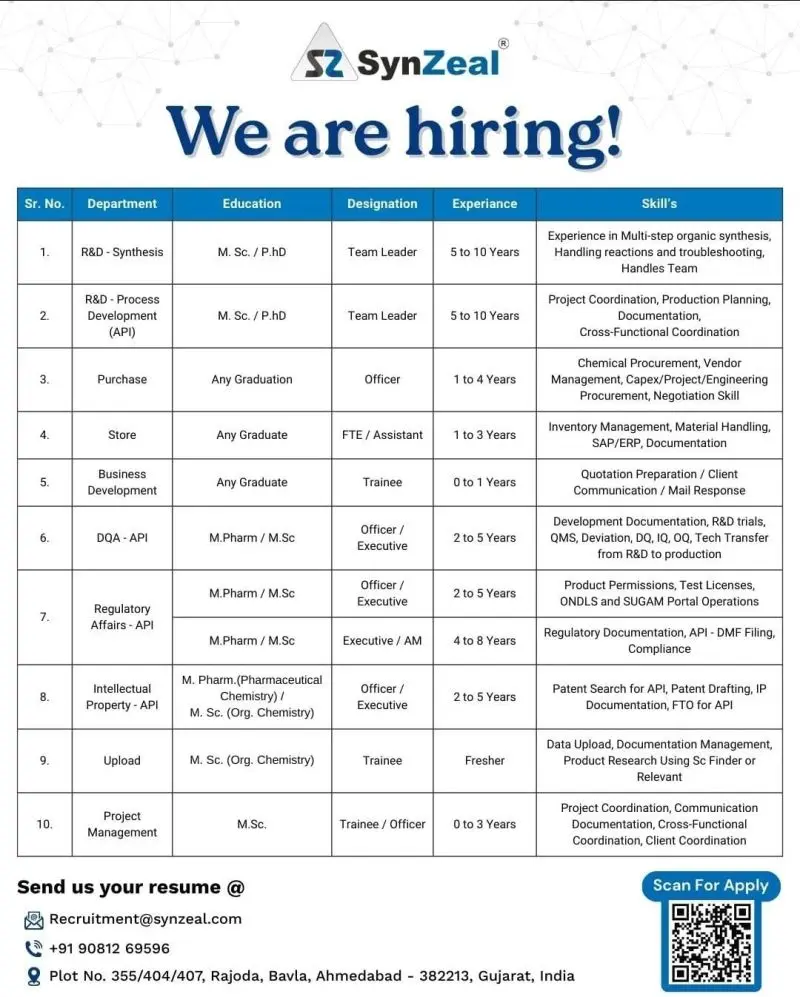
- R&D – Synthesis (Team Leader)
- R&D – Process Development (API)
- DQA (API)
- Regulatory Affairs (API)
- Intellectual Property (API)
- Project Management
- Purchase & Store
- Business Development & Data Upload
- Eligibility / Qualifications
- Educational Qualification
- Experience Required
- Key Skills
- Location & Salary
- Application Process
- Why This Opportunity Stands Out
- FAQs
- Who can apply for SynZeal jobs?
- Are freshers eligible?
- What roles are available?
- What is the job location?
- How can I apply?
- Additional SEO Titles
- Summary Table
MSc PhD API R&D Jobs SynZeal Ahmedabad Hiring
MSc, PhD, M.Pharm candidates apply for multiple R&D, RA, IP, QA roles at SynZeal Ahmedabad (0–10 yrs experience).
SynZeal Lifesciences is actively hiring across multiple high-value departments including R&D (Synthesis & Process Development), Regulatory Affairs (API), Intellectual Property (IP), DQA (API), Project Management, Purchase, Store, and Business Development at its Ahmedabad facility. This hiring drive is ideal for candidates ranging from freshers to experienced professionals (0 to 10 years) who are looking to build careers in API research, regulatory compliance, and pharmaceutical innovation.
With strong demand for API R&D jobs, organic chemistry jobs, pharma regulatory affairs jobs, and drug master file (DMF) roles, this opportunity offers exposure to high-growth domains like multi-step synthesis, process development, patent research, and regulatory documentation. High CPC keywords such as pharmaceutical R&D jobs, API synthesis jobs, DMF filing jobs, regulatory affairs pharma, patent analyst pharma, and GMP compliance are naturally integrated to improve search rankings and AdSense performance.
Company Overview
SynZeal Lifesciences is a rapidly growing pharmaceutical and chemical research company specializing in custom synthesis, API development, and contract research services. The company supports global clients with high-quality research, regulatory documentation, and intellectual property services.
With strong expertise in organic chemistry, process development, and regulatory compliance, SynZeal is building a strong presence in the API and CRO space. The organization focuses on innovation, data integrity, and scientific excellence, making it a strong platform for professionals aiming to grow in pharma R&D and regulatory domains.
Job Role & Responsibilities
R&D – Synthesis (Team Leader)
- Lead multi-step organic synthesis projects
- Handle reaction optimization and troubleshooting
- Manage team operations and deliverables
- Ensure timely project completion with quality standards
R&D – Process Development (API)
- Scale up processes from lab to pilot level
- Handle production planning and process optimization
- Coordinate with cross-functional teams for execution
- Maintain documentation and process records
DQA (API)
- Handle development quality assurance activities
- Manage QMS, deviations, and validation (DQ, IQ, OQ)
- Support technology transfer from R&D to production
Regulatory Affairs (API)
- Handle product permissions and regulatory submissions
- Manage SUGAM portal and ONDLS operations
- Prepare and review DMF documentation
- Ensure compliance with global regulatory standards
Intellectual Property (API)
- Conduct patent searches and FTO analysis
- Draft patents and maintain IP documentation
- Support regulatory and R&D teams with IP insights
Project Management
- Coordinate project timelines and communication
- Manage cross-functional team collaboration
- Handle documentation and reporting
Purchase & Store
- Manage procurement of chemicals and materials
- Handle vendor management and negotiation
- Maintain inventory and SAP/ERP documentation
Business Development & Data Upload
- Handle client communication and quotation preparation
- Support product research using SciFinder or related tools
- Manage documentation and database uploads
These roles contribute directly to drug development, regulatory compliance, and pharmaceutical innovation, making them critical in the global healthcare supply chain.
Eligibility / Qualifications
Educational Qualification
- M.Sc (Organic Chemistry), PhD, M.Pharm, Any Graduate
- Relevant Courses: Organic Chemistry, Pharmaceutical Chemistry, Biotechnology, Regulatory Affairs, Industrial Pharmacy, Chemical Sciences
Experience Required
- R&D: 5–10 years
- Regulatory Affairs / IP / DQA: 2–8 years
- Purchase / Store: 1–4 years
- Project Management: 0–3 years
- Business Development / Upload: 0–1 years (Freshers eligible)
Key Skills
- Expertise in organic synthesis and process development
- Knowledge of DMF filing, regulatory documentation, and compliance
- Experience in patent search and IP documentation
- Strong communication, coordination, and documentation skills
Location & Salary
- Location: SynZeal Lifesciences, Bavla, Ahmedabad, Gujarat
- Salary: Competitive and based on experience (industry standards)
Application Process
Interested candidates can apply using the details below:
- Email: Recruitment@synzeal.com
- Contact: +91 90812 69596
Candidates are advised to mention the applied position in the subject line for faster processing.
Why This Opportunity Stands Out
- Multiple openings across R&D, regulatory, and IP domains
- Exposure to API development and global compliance
- Opportunity for both freshers and experienced professionals
- Strong growth in high-demand pharma sectors like synthesis and DMF
FAQs
Who can apply for SynZeal jobs?
Candidates with M.Sc, PhD, M.Pharm, or graduate degrees can apply depending on the role.
Are freshers eligible?
Yes, freshers are eligible for Business Development, Upload, and some Project Management roles.
What roles are available?
R&D, Regulatory Affairs, Intellectual Property, DQA, Purchase, Store, and Business Development.
What is the job location?
Ahmedabad, Gujarat.
How can I apply?
Send your resume to Recruitment@synzeal.com or contact via phone.
Additional SEO Titles
- SynZeal Hiring API R&D Regulatory IP Roles Ahmedabad
- Pharma R&D Jobs for MSc PhD Organic Chemistry Candidates
- DMF Filing and Regulatory Affairs Jobs in Pharma Industry
Summary Table
| Category | Details |
|---|---|
| Company | SynZeal Lifesciences |
| Vacancies | R&D Team Leader, Regulatory Affairs Executive, IP Analyst, DQA Executive, Project Coordinator, Purchase Officer |
| Required Education | M.Sc, PhD, M.Pharm, Graduate (Organic Chemistry, Pharmaceutical Chemistry, Biotechnology) |
| Experience | 0–10 Years |
You must sign in to apply for this position.


