Tata Consultancy Walk-in Medical Reviewer, PV Case Processor, Safety Scientist & Statistical Programmer

- Company Overview
- Job Role & Responsibilities
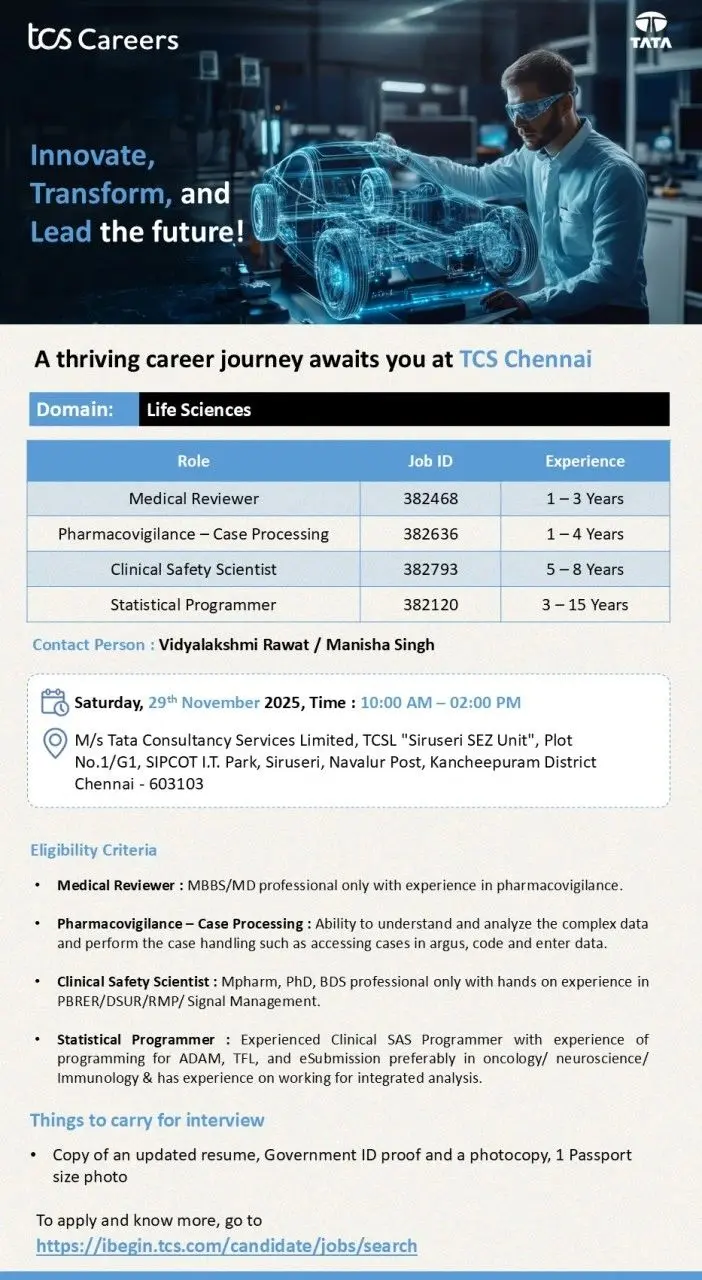
- Medical Reviewer – Job ID 382468 (1–3 Years)
- Pharmacovigilance – Case Processing – Job ID 382636 (1–4 Years)
- Clinical Safety Scientist – Job ID 382793 (5–8 Years)
- Statistical Programmer – Job ID 382120 (3–15 Years)
- Eligibility / Qualifications
- Things to Carry for Interview
- Location & Schedule
- Application Process
- FAQs
- Who can apply for the Medical Reviewer role?
- What systems are used for PV Case Processing?
- What experience is required for Clinical Safety Scientist?
- What skills are needed for Statistical Programming?
- Can candidates with CRO experience apply?
- Summary Table
Life Sciences Roles at TCS – Medical Reviewer & PV, Chennai
TCS Chennai hiring Medical Reviewer, PV Case Processor, Safety Scientist & Statistical Programmer. Walk-in on 29 Nov 2025.
TCS is hiring experienced Life Sciences professionals for key roles across Medical Review, Pharmacovigilance, Clinical Safety and Statistical Programming. The walk-in drive in Chennai offers strong career growth for candidates with domain expertise and hands-on exposure to global safety and clinical deliverables. These positions suit professionals aiming to work in a structured, high-compliance environment that supports global healthcare projects.
Company Overview
Tata Consultancy Services (TCS) is one of the world’s leading IT and consulting organizations with a strong Life Sciences and Healthcare practice. TCS supports global pharmaceutical, biotech and CRO partners across drug safety, clinical development, regulatory operations and biometrics. With advanced analytics, automation and domain-driven solutions, TCS is recognised for delivering high-quality scientific and technology services. Its Chennai SEZ unit is a major delivery centre for safety, PV operations and clinical analytics.
Job Role & Responsibilities
Medical Reviewer – Job ID 382468 (1–3 Years)
- Review Individual Case Safety Reports (ICSRs) for clinical and post-marketing safety.
- Perform medical assessment, narrative review and seriousness classification.
- Ensure compliance with global PV guidelines and timelines.
- Validate coding, causality and MedDRA terms.
Pharmacovigilance – Case Processing – Job ID 382636 (1–4 Years)
- Perform case intake, triage, data entry and medical coding in systems such as Argus.
- Retrieve, assess and process adverse event reports from multiple sources.
- Analyze clinical and safety data, maintain accuracy and completeness of ICSRs.
- Follow SOPs, compliance standards and global reporting expectations.
Clinical Safety Scientist – Job ID 382793 (5–8 Years)
- Prepare and review PBRERs, DSURs, Risk Management Plans and safety sections of regulatory documents.
- Conduct signal detection, evaluation and safety surveillance.
- Work with cross-functional teams on safety strategy and regulatory reporting.
- Maintain safety intelligence, literature review and benefit-risk evaluations.
Statistical Programmer – Job ID 382120 (3–15 Years)
- Develop and validate SAS programs for ADaM datasets, TFLs and e-submissions.
- Support integrated analysis, regulatory submissions and ISS/ISE packages.
- Work on studies in oncology, immunology or neuroscience.
- Ensure adherence to CDISC standards, QA processes and statistical programming practices.
Eligibility / Qualifications
- Medical Reviewer: MBBS/MD with pharmacovigilance experience.
- PV Case Processing: Graduates with strong PV knowledge and Argus case-handling exposure.
- Clinical Safety Scientist: M.Pharm, PhD, BDS with strong experience in PBRER/DSUR/RMP/Signal Management.
- Statistical Programmer: Clinical SAS Programmer with ADaM, TFL, eSubmission experience.
Relevant Courses (comma-separated): MBBS, MD, M.Pharm, B.Pharm, BDS, PhD Life Sciences, Clinical SAS Programming, CDISC ADaM training, PV Argus certification, Clinical Research courses, Drug Safety training modules.
Things to Carry for Interview
- Updated resume.
- Government ID proof + photocopy.
- One passport-size photograph.
Location & Schedule
- Walk-In Date: Saturday, 29 November 2025
- Time: 10:00 AM – 02:00 PM
- Venue: TCSL Siruseri SEZ Unit, Plot No. 1/G1, SIPCOT IT Park, Siruseri, Navalur Post, Kancheepuram District, Chennai – 603103.
Application Process
To apply online and view detailed job descriptions:
https://ibegin.tcs.com/candidate/jobs/search
You may also walk in directly with the required documents.
FAQs
Who can apply for the Medical Reviewer role?
Only MBBS/MD professionals with PV experience.
What systems are used for PV Case Processing?
Argus and other safety databases.
What experience is required for Clinical Safety Scientist?
5–8 years with PBRER/DSUR/RMP/Signal Management.
What skills are needed for Statistical Programming?
Strong SAS programming, ADaM, TFL, eSubmission and CDISC experience.
Can candidates with CRO experience apply?
Yes. CRO, pharma or global safety experience is preferred.
Summary Table
| Category | Details |
|---|---|
| Company | Tata Consultancy Services (TCS) |
| Vacancies | Medical Reviewer, PV Case Processing, Clinical Safety Scientist, Statistical Programmer |
| Required Education | MBBS/MD, M.Pharm, BDS, PhD, SAS/Clinical Research Certifications |
| Experience | 1–15 years depending on the role |