JDM Research Hiring Phys-Chem, R&D, IPR, and Toxicology

- M.Sc & M.Pharm Openings at JDM Research – Vadodara
- Company Overview
- Job Role & Responsibilities
- Phys-Chem Department
- Research & Development – Synthesis
- Research & Development – Analytical
- Intellectual Property Rights (IPR)
- Toxicology Department
- Eligibility / Qualifications
- Location & Salary
- Application Process
- FAQs
- Summary Table
M.Sc & M.Pharm Openings at JDM Research – Vadodara
JDM Research hiring M.Sc/M.Pharm professionals for Phys-Chem, R&D, IPR, and Toxicology roles. Multiple vacancies in Vadodara.
JDM Scientific Research Organisation Pvt. Ltd. is hiring across Phys-Chem, R&D, IPR, and Toxicology departments. These openings suit candidates with hands-on experience in GLP labs, OECD guidelines, method development, synthesis, toxicology studies, and intellectual property rights. The organisation prefers candidates who can work in a regulated CRO environment and follow global compliance standards. Applicants must mention the relevant Job Code in the email subject line.
Company Overview
JDM Scientific Research Organisation is a fully integrated Contract Research Organisation (CRO) accredited with ISO 17025, GLP, and DSIR certifications. Located in Vadodara, the company specialises in phys-chem studies, toxicology research, synthesis, analytical development, regulatory documentation, and IP management. JDM supports global clients through validated processes aligned with OECD, OPPTS, SANCO, ABNT, CIPAC, and other international guidelines. Its infrastructure includes advanced laboratories, controlled environments, and expert scientific teams.
Job Role & Responsibilities
Phys-Chem Department
Officer / Executive / Senior Executive – GLP Physical Chemical Testing (Job Code: PHYS-CHEM)
- Conduct five-batch and phys-chem studies as per OECD, OPPTS, SANCO, ABNT, CIPAC guidelines.
- Function as Study Director for physico-chemical studies.
- Prepare and review study plans, raw data, and final study reports.
- Ensure GLP compliance and accurate documentation.
Research & Development – Synthesis
R&D Officer / Executive – Synthesis (Job Code: SYN)
- Execute organic synthesis experiments at laboratory scale.
- Conduct literature search and develop synthetic pathways.
- Support technology transfer to pilot and commercial-scale facilities.
- Prepare experiment documentation and maintain compliance.
Research & Development – Analytical
R&D Officer / Executive – Analytical (Job Code: ADL)
- Perform analytical method development and validation using HPLC and GC.
- Support stability studies, impurity analysis, and analytical troubleshooting.
- Prepare protocols, reports, and analytical documentation.
- Ensure adherence to GLP and OECD guidelines.
Intellectual Property Rights (IPR)
Executive / Senior Executive – IPR (Job Code: IPR)
- Perform detailed literature searches and patentability assessments.
- Draft and file patents, trademarks, copyrights, and design applications.
- Support litigation and documentation activities.
- Maintain IPR records and coordinate with R&D for technical inputs.
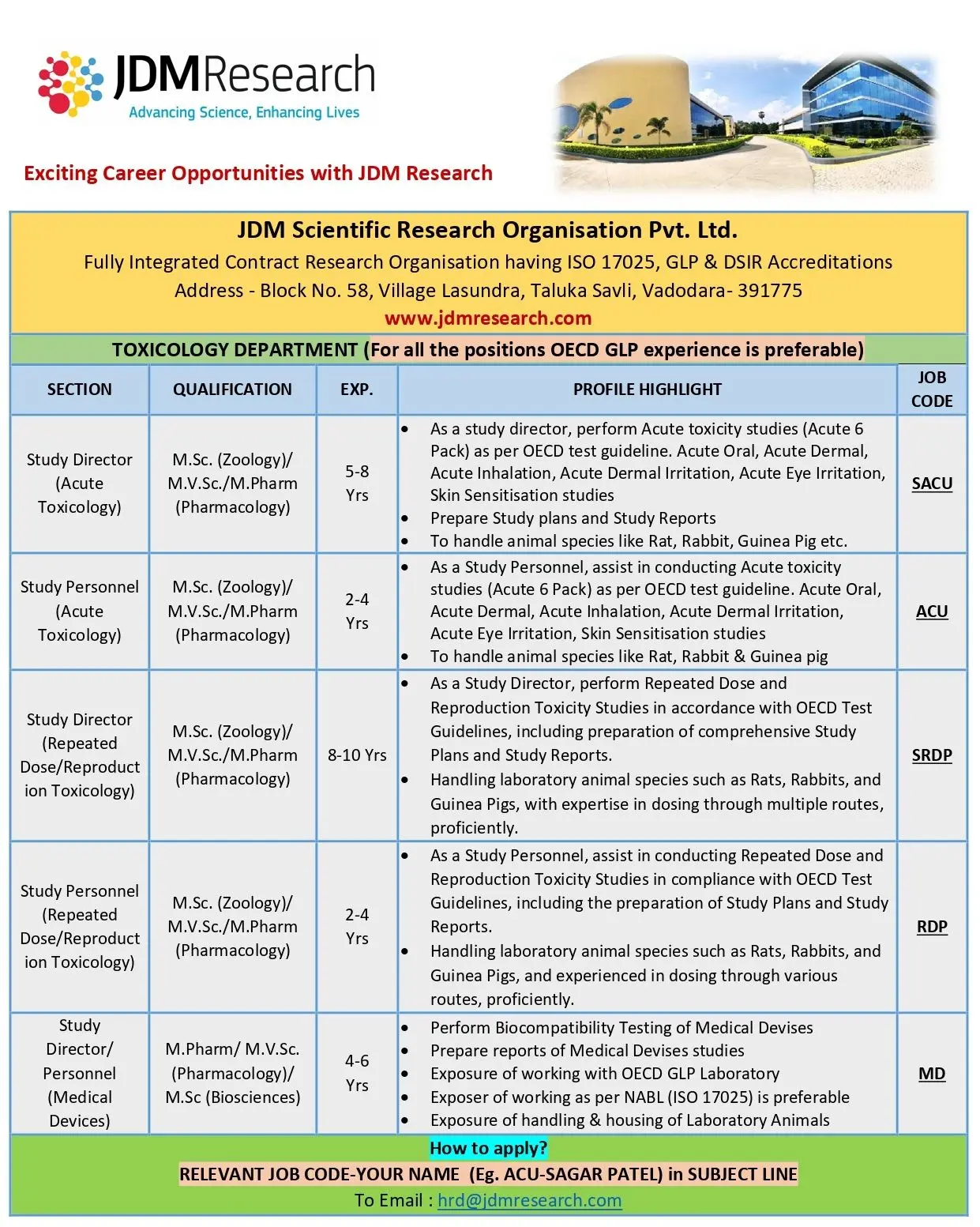
Toxicology Department
(GLP experience preferred for all positions)
Acute Toxicology
Study Director – Acute Toxicology (Job Code: SACU)
- Conduct acute toxicity studies (Acute 6 Pack) as per OECD guidelines.
- Prepare study plans, raw data summaries, and final reports.
- Handle laboratory species such as Rats, Rabbits, and Guinea Pigs.
Study Personnel – Acute Toxicology (Job Code: ACU)
- Assist in conducting acute toxicity studies.
- Support data recording, dosing, and handling of animal species.
- Work under Study Director guidance.
Repeated Dose / Reproduction Toxicology
Study Director – Repeated Dose/Reproduction Toxicology (Job Code: SRDP)
- Lead repeated dose and reproductive toxicity studies.
- Draft study plans and final reports as per OECD guidelines.
- Manage animal handling and dosing across species.
Study Personnel – Repeated Dose/Reproduction Toxicology (Job Code: RDP)
- Assist in study execution, dosing, observations, and documentation.
- Support report generation and study plan preparation.
Medical Devices
Study Director / Study Personnel – Medical Devices (Job Code: MD)
- Perform biocompatibility studies of medical devices.
- Prepare study plans and final reports.
- Ensure compliance with OECD GLP and ISO 17025 expectations.
- Manage handling and housing of laboratory animals.
Eligibility / Qualifications
- Phys-Chem: M.Sc Analytical Chemistry, 2–6 years
- R&D – Synthesis: M.Sc Organic Chemistry, 2–6 years
- R&D – Analytical: M.Sc Analytical/Organic Chemistry, 2–6 years
- IPR: M.Sc Chemistry or M.Pharm with 5–8 years IPR experience
- Toxicology Study Directors: M.Sc Zoology / M.V.Sc / M.Pharm Pharmacology with 5–10 years experience
- Toxicology Study Personnel: Same qualifications with 2–4 years experience
- Medical Devices: M.Pharm/M.V.Sc/M.Sc Biosciences with 4–6 years experience
Relevant courses (comma-separated): M.Sc Analytical Chemistry, M.Sc Organic Chemistry, M.Sc Zoology, M.Sc Biosciences, M.Pharm Pharmacology, PG Diploma in Toxicology, Certificate in GLP, Certificate in ISO 17025, PG Diploma in IPR.
Location & Salary
- Work Location: Block No. 58, Village Lasundra, Savli, Vadodara – 391775
- Salary: Competitive and based on experience and job level.
Application Process
Candidates must mention the correct Job Code in the subject line as:
– (e.g., SYN – Sagar Patel)
Send your CV to:
hrd@jdmresearch.com
Shortlisted candidates will be contacted for further rounds.
FAQs
1. Is GLP experience mandatory?
Preferred for most roles, strongly preferred for toxicology and phys-chem positions.
2. What is the experience range?
Roles require 2–10 years depending on the position.
3. Can freshers apply?
No. Only experienced candidates will be considered.
4. Are these full-time on-site roles?
Yes. All positions require on-site presence at the Vadodara facility.
5. What should be included in the application email?
Mention the Job Code and your full name in the subject line.
Summary Table
| Category | Details |
|---|---|
| Company | JDM Scientific Research Organisation Pvt. Ltd. |
| Vacancies | Phys-Chem, R&D (Synthesis/Analytical), IPR, Toxicology (Acute, Repeated Dose, Medical Devices) |
| Required Education | M.Sc Analytical/Organic Chemistry, M.Pharm Pharmacology, M.Sc Zoology, M.Sc Biosciences |
| Experience | 2–10 years depending on department & Job Code |

You must sign in to apply for this position.


