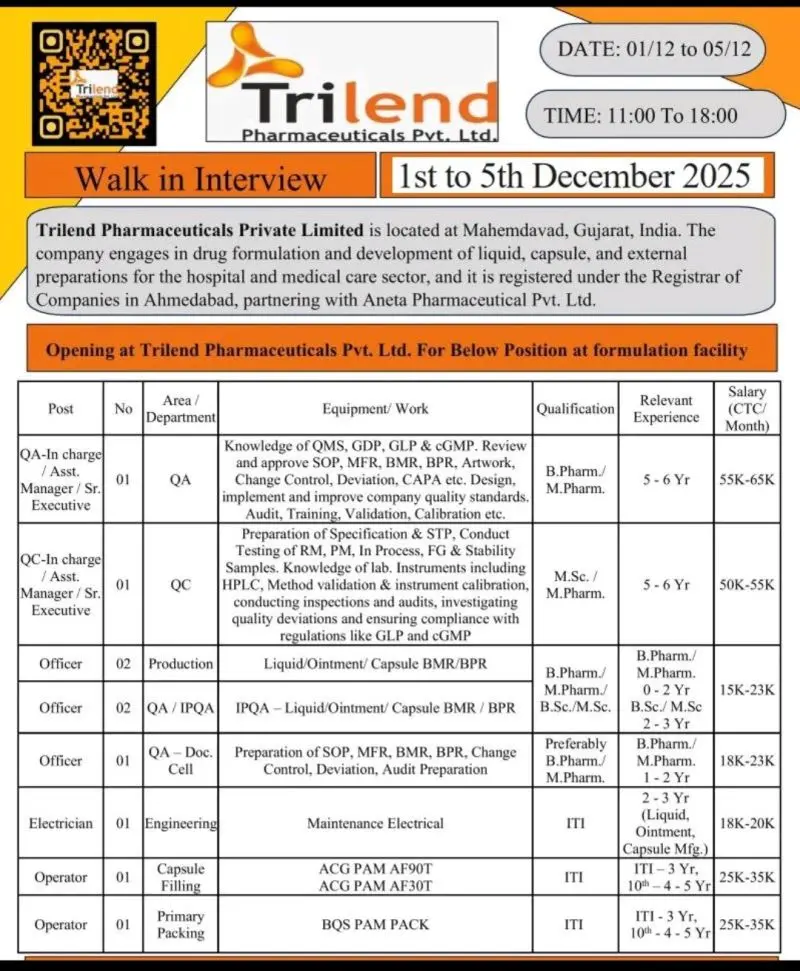
Trilend Walk-in QA, QC, Production, IPQA, Engineering

- B.Pharm QA QC Production Openings – Trilend Gujarat
- Company Overview
- Job Role & Responsibilities
- QA – In‑Charge / Assistant Manager / Senior Executive
- QC – In‑Charge / Assistant Manager / Senior Executive
- Production – Officer (Liquid / Ointment / Capsule)
- QA/IPQA – Officer
- QA – Documentation Cell (Officer)
- Engineering – Electrician
- Capsule Filling – Operator
- Primary Packing – Operator
- Eligibility / Qualifications
- Location & Salary
- Application Process
- FAQs
- Summary Table
B.Pharm QA QC Production Openings – Trilend Gujarat
Walk-in for QA, QC, Production, IPQA, Engineering roles at Trilend Pharmaceuticals, Gujarat. Hiring 0–6 yrs experienced candidates.
Trilend Pharmaceuticals Pvt. Ltd. is hiring across QA, QC, Production, IPQA, and Engineering functions for its formulation facility in Mahemdavad, Gujarat. These openings suit candidates with hands-on exposure to liquid, ointment, and capsule dosage forms, as well as strong understanding of QMS, cGMP, GLP and analytical operations. The walk-in drive runs from 1st to 5th December 2025.
Company Overview
Trilend Pharmaceuticals is a formulation development and manufacturing company based in Mahemdavad, Gujarat. The organisation focuses on liquid, capsule, and external preparations for hospital and medical care sectors. The facility operates under cGMP, QMS, and GLP-aligned systems and works in collaboration with Aneta Pharmaceuticals Pvt. Ltd., supporting end‑to‑end dosage form development, quality operations, and regulated documentation.
Job Role & Responsibilities
QA – In‑Charge / Assistant Manager / Senior Executive
- Oversee QMS, GDP, GLP, and cGMP compliance.
- Review and approve SOPs, MFR, BMR, BPR, Artwork, Change Control, Deviations, and CAPA.
- Manage internal audits, validation, qualification, calibration, and training activities.
- Implement and enhance company-wide quality standards.
QC – In‑Charge / Assistant Manager / Senior Executive
- Prepare Specifications and STPs.
- Conduct RM, PM, In-process, Finished Goods, and Stability testing.
- Operate and troubleshoot HPLC and other analytical instruments.
- Support method validation, instrument calibration, investigations, and audit readiness.
Production – Officer (Liquid / Ointment / Capsule)
- Handle BMR/BPR and shop‑floor operations for liquid, ointment, and capsule dosage forms.
- Ensure smooth batch execution, documentation, and in‑process compliance.
- Support equipment handling, material management, and basic troubleshooting.
QA/IPQA – Officer
- Perform line clearance, sampling, and IPQA checks for liquid, ointment, and capsule lines.
- Review BMR/BPR and ensure adherence to cGMP and quality expectations.
- Monitor in‑process controls and documentation accuracy.
QA – Documentation Cell (Officer)
- Prepare and control SOPs, MFR, BMR, BPR.
- Manage deviation logs, audit preparation, and document change control.
- Coordinate with QA leads for record compliance.
Engineering – Electrician
- Responsible for electrical maintenance of liquid, ointment, and capsule manufacturing areas.
- Perform preventive maintenance, troubleshooting, and breakdown support.
Capsule Filling – Operator
- Operate ACG PAM AF90T / AF30T capsule filling machines.
- Conduct equipment setup, changeover, cleaning, and documentation.
- Monitor capsule weight, locking, polishing, and output.
Primary Packing – Operator
- Operate BQS PAM PACK line equipment.
- Execute packing operations as per batch requirements.
- Maintain GMP and safety compliance.
Eligibility / Qualifications
- QA In‑Charge / AM / Sr. Executive: B.Pharm / M.Pharm, 5–6 yrs
- QC In‑Charge / AM / Sr. Executive: M.Sc / M.Pharm, 5–6 yrs
- Production Officer: B.Pharm/M.Pharm (0–2 yrs) or B.Sc/M.Sc (2–3 yrs)
- QA/IPQA Officer: B.Pharm/M.Pharm, 1–2 yrs
- QA Documentation: B.Pharm/M.Pharm, 1–3 yrs
- Electrician: ITI with 2–3 yrs in liquid/ointment/capsule manufacturing plants
- Capsule Filling Operator: ITI (3 yrs) or 10th pass with 4–5 yrs experience
- Primary Packing Operator: ITI (3 yrs) or 10th pass with 4–5 yrs experience
Relevant courses (comma‑separated): B.Pharm, M.Pharm, B.Sc Chemistry, M.Sc Chemistry, M.Sc Pharmaceutical Sciences, ITI Electrical, ITI Mechanical, Certificate in GMP, PG Diploma in QA.
Location & Salary
- Location: Trilend Pharmaceuticals Pvt. Ltd., Mahemdavad, Gujarat.
- Salary Range: ₹15,000 – ₹65,000 per month depending on role and experience.
- Walk‑in Dates: 1st to 5th December 2025, 11:00 AM – 06:00 PM.
Application Process
Attend the walk‑in directly with updated CV, experience certificates, and ID proof.
Walk‑in Address: Trilend Pharmaceuticals Pvt. Ltd., Mahemdavad, Gujarat.
If unable to attend, candidates may request the official HR email at the venue or company website.
FAQs
1. Can freshers apply?
Yes. Production (B.Pharm/M.Pharm) and specific officer roles accept freshers.
2. Are multiple departments hiring?
Yes. QA, QC, Production, IPQA, Engineering, and Capsule/Primary Packing.
3. Is GMP experience required?
Preferred, especially for QA/QC/IPQA roles.
4. Will there be on‑site tests or interviews?
Likely. Candidates should be prepared for technical discussions.
5. What should candidates bring?
CV, certificates, passport‑size photos, and any compliance or training documents.
Summary Table
| Category | Details |
|---|---|
| Company | Trilend Pharmaceuticals Pvt. Ltd. |
| Vacancies | QA, QC, Production, IPQA, Documentation, Engineering, Operators |
| Required Education | B.Pharm, M.Pharm, B.Sc, M.Sc, ITI |
| Experience | 0–6 years depending on role |