Innoxel Hiring QC, QA, Microbiology, Production, Engineering

- Company Overview
- Job Role & Responsibilities
- Quality Control (QC – Analytical)
- Sterile / Liquid Oral Manufacturing
- QC Microbiology
- Quality Assurance (QA)
- Warehouse
- Validation
- Engineering & Maintenance
- Project Management
- Eligibility / Qualifications
- Required Education
- Relevant Courses
- Experience
- Skills Required
- Location & Salary
- Job Location
- Salary
- Application Process
- How to Apply
- Why You Should Apply
- FAQs
- Who can apply for these roles?
- Is USFDA experience mandatory?
- Which departments are hiring?
- How can I apply?
- What qualifications are required?
- Summary Table
Innoxel Lifesciences Hiring 2026 | QC, QA, Microbiology, Production, Engineering Jobs Across Multiple Departments
Innoxel Lifesciences is actively hiring experienced professionals across Quality Control, Quality Assurance, Microbiology, Manufacturing, Engineering, Warehouse, Validation, and Project Management departments. This recruitment drive offers multiple vacancies for Analysts, Officers, Executives, Operators, and Supervisors with experience ranging from 1 to 10 years in sterile USFDA-regulated pharmaceutical environments.
This is a strong opportunity for candidates looking to advance their careers in pharmaceutical manufacturing, regulatory compliance, analytical development, and sterile injectable operations within a globally compliant organization.
Company Overview
Innoxel Lifesciences is a growing pharmaceutical company focused on delivering high-quality formulations with a strong emphasis on sterile manufacturing and regulatory compliance. The company operates with adherence to USFDA standards and global quality benchmarks, making it a trusted name in pharmaceutical manufacturing and export markets.
With a strong infrastructure, advanced technology platforms, and a compliance-driven culture, Innoxel Lifesciences provides professionals with exposure to global regulatory practices, making it an ideal workplace for career growth in pharma production, quality systems, and engineering operations.
Job Role & Responsibilities
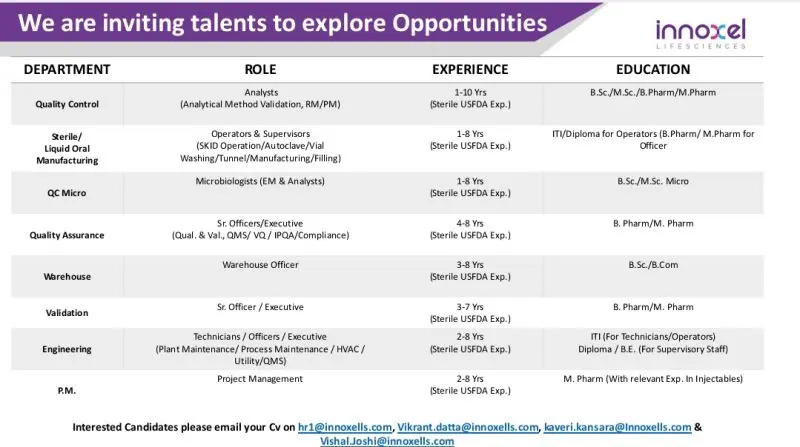
Quality Control (QC – Analytical)
- Perform analytical method validation and testing of raw materials and packaging materials
- Handle analytical instruments such as HPLC, GC, and other lab systems
- Maintain GLP-compliant documentation and reporting
- Ensure adherence to regulatory guidelines and quality standards
Sterile / Liquid Oral Manufacturing
- Operate manufacturing equipment including autoclave, vial washing, tunnel, and filling lines
- Handle SKID operations and sterile injectable production processes
- Maintain batch manufacturing records and ensure GMP compliance
- Supervise production activities and ensure quality output
QC Microbiology
- Conduct environmental monitoring (EM) and microbiological testing
- Perform microbial analysis and laboratory documentation
- Ensure compliance with sterility and contamination control protocols
Quality Assurance (QA)
- Handle qualification, validation, QMS, IPQA, and compliance activities
- Review batch records and ensure GMP adherence
- Support audits and regulatory inspections
Warehouse
- Manage inventory, material handling, and documentation
- Ensure proper storage and tracking of pharmaceutical materials
- Maintain compliance with warehouse SOPs and regulatory guidelines
Validation
- Execute validation protocols for equipment and processes
- Prepare validation reports and ensure regulatory compliance
Engineering & Maintenance
- Perform plant and process maintenance including HVAC and utility systems
- Handle calibration and preventive maintenance activities
- Ensure smooth functioning of pharmaceutical equipment
Project Management
- Manage pharmaceutical projects related to sterile injectables
- Coordinate cross-functional teams for project execution
- Ensure timelines, compliance, and documentation standards are met
Eligibility / Qualifications
Required Education
B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma, B.E., B.Com
Relevant Courses
Pharmaceutical Sciences, Microbiology, Biotechnology, Chemistry, Industrial Pharmacy, Mechanical Engineering, Instrumentation, Quality Assurance, Regulatory Affairs, Production Technology
Experience
- 1 to 10 years of experience in pharmaceutical industry
- Mandatory experience in sterile USFDA-regulated environment
Skills Required
- Strong knowledge of GMP, GLP, and regulatory compliance
- Hands-on experience with analytical instruments and manufacturing systems
- Good documentation and audit handling skills
- Ability to work in a regulated pharma environment
Location & Salary
Job Location
- Location not explicitly mentioned (based on company operations and communication during hiring process)
Salary
- Competitive salary package based on experience and role
- Performance-driven growth and career advancement opportunities
Application Process
How to Apply
Interested candidates can send their updated resumes to the following email IDs:
- hr1@innoxells.com
- Vikrant.datta@innoxells.com
- kaveri.kansara@innoxells.com
- Vishal Joshi@innoxells.com
Candidates are advised to mention the position applied for in the subject line for faster processing.
Why You Should Apply
- Opportunity to work in USFDA-regulated sterile pharmaceutical environment
- Exposure to global compliance standards and regulatory practices
- Multiple department openings offering career flexibility
- Strong career growth in pharma manufacturing, QA, QC, and engineering
- Hands-on experience with advanced pharmaceutical technologies
FAQs
Who can apply for these roles?
Candidates with relevant qualifications and 1 to 10 years of experience in pharmaceutical industries, especially sterile environments, can apply.
Is USFDA experience mandatory?
Yes, candidates with sterile USFDA experience are preferred for most roles.
Which departments are hiring?
Quality Control, Quality Assurance, Microbiology, Manufacturing, Engineering, Warehouse, Validation, and Project Management.
How can I apply?
You can apply by sending your CV to the official email IDs mentioned above.
What qualifications are required?
Candidates with B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma, B.E., or related qualifications are eligible based on role.
Summary Table
| Company | Innoxel Lifesciences |
|---|---|
| Vacancies | QC Analyst, QA Officer/Executive, Microbiologist, Production Operator/Supervisor, Engineering Technician/Officer, Warehouse Officer, Validation Executive, Project Manager |
| Required Education | B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma, B.E., B.Com |
| Experience | 1–10 Years |
You must sign in to apply for this position.


