Aneta Walk-In ADL ,F&D ,QC ,RA

- Company Overview
- Job Role & Responsibilities
- Analytical R&D (ADL)
- Formulation & Development (F&D)
- Regulatory Affairs
- Quality Control (HPLC Analyst)
- Eligibility / Qualifications
- Required Education:
- Experience:
- Key Skills:
- Location & Salary
- Job Location:
- Salary:
- Application Process
- Walk-in Details:
- Documents to Carry:
- Contact:
- Why This Role Matters in Pharma Industry
- Frequently Asked Questions (FAQs)
- 1. Who can attend this walk-in drive?
- 2. Are freshers eligible?
- 3. What is the selection process?
- 4. Is prior pharma experience mandatory?
- 5. What skills are important?
- Summary
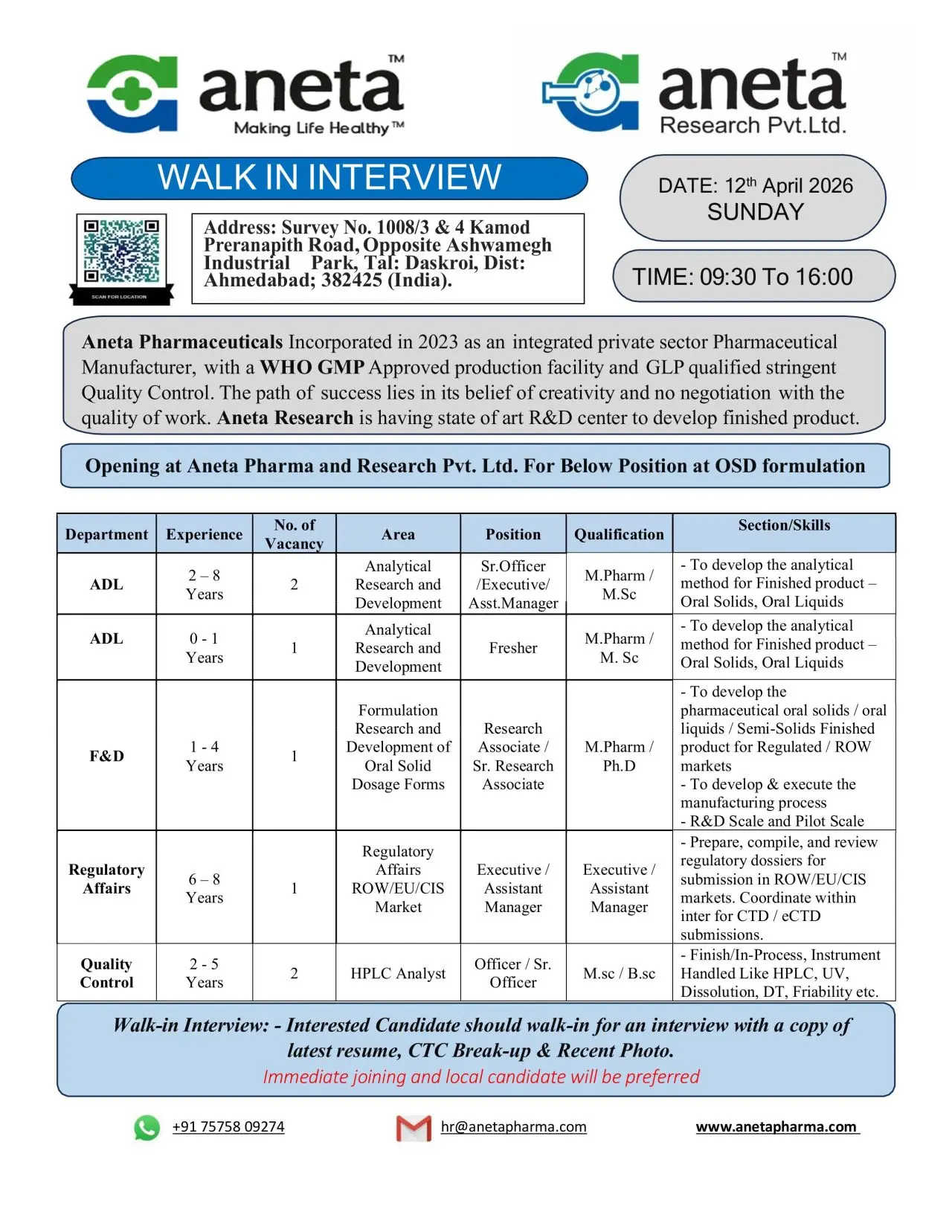
Aneta Research Pvt. Ltd. is conducting a walk-in interview for multiple vacancies in Analytical R&D (ADL), Formulation & Development (F&D), Regulatory Affairs, and Quality Control departments. A total of 6+ openings are available across fresher and experienced roles, making this an excellent opportunity for both fresh graduates and professionals looking to advance in pharmaceutical research and development. The walk-in drive will be held on 12th April 2026 in Ahmedabad, Gujarat, for positions based in OSD formulation.
Company Overview
Aneta Pharma and Research Pvt. Ltd., established in 2023, is a fast-growing pharmaceutical manufacturing company with a strong focus on quality-driven production and innovation. The company operates with a WHO-GMP approved manufacturing facility and follows stringent GLP standards in its quality control systems.
With a state-of-the-art R&D center, Aneta focuses on developing high-quality finished dosage forms for regulated and ROW markets. The organization is committed to innovation, compliance, and delivering safe pharmaceutical products that support global healthcare systems.
Working at Aneta Pharma offers professionals exposure to advanced analytical techniques, formulation development, regulatory submissions, and global pharmaceutical standards.
Job Role & Responsibilities
Analytical R&D (ADL)
- Develop and validate analytical methods for oral solid and liquid dosage forms
- Perform analytical testing using advanced instruments
- Support stability studies and method transfer activities
Formulation & Development (F&D)
- Develop oral solid, liquid, and semi-solid formulations for regulated markets
- Execute R&D scale and pilot scale batches
- Support technology transfer and process optimization
Regulatory Affairs
- Prepare and review regulatory dossiers for ROW, EU, and CIS markets
- Handle CTD/eCTD submissions and documentation
- Coordinate with internal teams for regulatory compliance
Quality Control (HPLC Analyst)
- Perform analysis of raw materials, intermediates, and finished products
- Operate instruments like HPLC, UV, Dissolution apparatus, and related equipment
- Maintain GLP compliance and documentation
Eligibility / Qualifications
Required Education:
M.Pharm, M.Sc, B.Sc, Ph.D (Pharmaceutical Sciences, Chemistry, Analytical Chemistry, Organic Chemistry, Pharmaceutics)
Experience:
- Freshers: 0–1 year (Analytical R&D)
- Experienced roles: 1–8 years depending on department
Key Skills:
- Knowledge of analytical method development and validation
- Experience in formulation development and process optimization
- Understanding of regulatory submissions and global compliance
- Hands-on experience with HPLC and analytical instruments
Location & Salary
Job Location:
Ahmedabad, Gujarat
Salary:
Salary will be offered as per industry standards based on experience, technical expertise, and role. Candidates with strong R&D and regulatory experience can expect competitive compensation packages.
Application Process
Eligible candidates can directly attend the walk-in interview with required documents.
Walk-in Details:
- Date: 12th April 2026 (Sunday)
- Time: 09:30 AM to 04:00 PM
- Venue: Survey No. 1008/3 & 4, Kamod Preranapith Road, Opp. Ashwamegh Industrial Park, Daskroi, Ahmedabad – 382425
Documents to Carry:
- Updated resume
- Recent passport-size photograph
- CTC breakup details
Contact:
- Email: Mhr@anetapharma.com
- Phone: +91 7575809274
- Website: www.anetapharma.com
Immediate joiners and local candidates will be given preference during the selection process.
Why This Role Matters in Pharma Industry
Roles in Analytical R&D, Formulation Development, and Regulatory Affairs are critical to the pharmaceutical lifecycle. These functions ensure drug quality, stability, regulatory compliance, and successful product launches across global markets.
By joining Aneta Pharma, candidates will contribute to the development of safe and effective medicines, directly impacting patient care and healthcare innovation. The exposure to WHO-GMP standards and international regulatory practices makes this opportunity valuable for long-term career growth.
Frequently Asked Questions (FAQs)
1. Who can attend this walk-in drive?
Candidates with M.Pharm, M.Sc, B.Sc, or Ph.D qualifications with relevant experience or freshers in ADL can attend.
2. Are freshers eligible?
Yes, freshers are eligible for Analytical R&D roles.
3. What is the selection process?
Walk-in interview followed by technical evaluation and HR discussion.
4. Is prior pharma experience mandatory?
Yes for experienced roles, but freshers can apply for entry-level ADL positions.
5. What skills are important?
Analytical skills, formulation knowledge, regulatory understanding, and instrument handling experience.
Summary
| Company | Aneta Pharma and Research Pvt. Ltd. |
|---|---|
| Vacancies | ADL, F&D, Regulatory Affairs, Quality Control (6+ Positions) |
| Required Education | M.Pharm, M.Sc, B.Sc, Ph.D (Pharma, Chemistry, Analytical, Organic) |
| Experience | 0–8 Years depending on role |