Cohance walk-in Production, QA, QC

- Company Overview
- Job Role & Responsibilities
- Production Roles
- Quality Control (QC)
- Quality Assurance (QA)
- Warehouse
- Engineering & Maintenance
- Eligibility / Qualifications
- Required Education:
- Relevant Courses:
- Experience Required:
- Skills Required:
- Location & Salary
- Application Process
- Walk-In Details:
- Interview Venue:
- Contact Details:
- Documents Required:
- Why Join Cohance Lifesciences?
- FAQs
- What experience is required?
- Are multiple roles available?
- Is formulation experience mandatory?
- What skills are required for QC roles?
- How to attend the interview?
- Final Thoughts
BPharm MSc Pharma Jobs 20+ Vacancies Hyderabad
B.Pharm, M.Pharm, MSc candidates apply for multiple vacancies in Production, QA, QC at Cohance Hyderabad. 3–6 yrs experience.
Cohance Lifesciences Limited is conducting a walk-in interview for multiple roles across Production, Quality Assurance, Quality Control, Warehouse, and Engineering departments. This hiring drive targets experienced pharmaceutical professionals with strong formulation and GMP exposure.
If you are working in OSD manufacturing, QC analysis, QA compliance, or engineering support in pharma plants, this is a direct opportunity to step into a regulated and growth-oriented organization.
Company Overview
Cohance Lifesciences Limited is a rapidly growing pharmaceutical organization focused on formulation development, manufacturing excellence, and regulatory compliance. The company operates multiple formulation plants and maintains strict adherence to GMP, quality systems, and global pharmaceutical standards.
With a strong emphasis on collaboration, operational excellence, and innovation, Cohance offers professionals a platform to work in regulated environments and contribute to high-quality medicine production.
Job Role & Responsibilities
The hiring spans across multiple departments. Each role is aligned with pharmaceutical manufacturing, analytical testing, and compliance-driven operations.
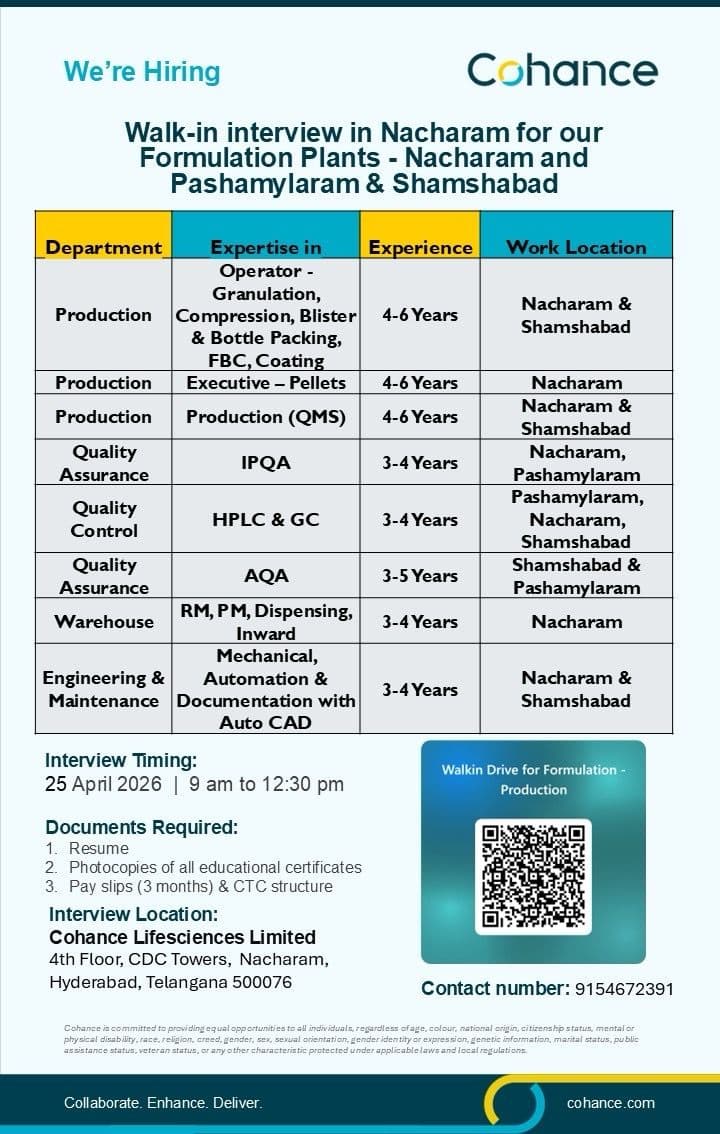
Production Roles
- Operate granulation, compression, coating, and packing equipment
- Handle pellet manufacturing and formulation processes
- Ensure batch manufacturing as per SOP and BMR
- Maintain documentation and process control
Quality Control (QC)
- Perform analysis using HPLC and GC
- Conduct raw material, in-process, and finished product testing
- Maintain analytical records and GMP compliance
Quality Assurance (QA)
- Handle IPQA and AQA activities
- Monitor in-process quality checks and compliance
- Manage deviation, CAPA, and documentation
Warehouse
- Manage raw material (RM) and packing material (PM)
- Handle dispensing, inward, and inventory control
Engineering & Maintenance
- Perform mechanical maintenance and automation support
- Handle documentation and AutoCAD-related tasks
- Ensure smooth plant operations and equipment reliability
These roles are critical in ensuring pharmaceutical product quality, regulatory compliance, and manufacturing efficiency.
Eligibility / Qualifications
Required Education:
- B.Pharm
- M.Pharm
- B.Sc / M.Sc
- Diploma / ITI (for technical roles)
Relevant Courses:
Pharmaceutical Manufacturing, GMP, GLP, HPLC Analysis, Quality Assurance, Tablet Compression, Coating Technology, Engineering Maintenance, AutoCAD, Warehouse Management
Experience Required:
- 3 to 6 years depending on role
Skills Required:
- Knowledge of GMP guidelines
- Hands-on experience in formulation plants
- Analytical skills for QC roles
- Documentation and compliance handling
Location & Salary
- Work Locations: Nacharam, Pashamylaram, Shamshabad (Hyderabad)
- Salary: Competitive and based on experience
- Additional Benefits: Exposure to regulated pharma environments and career growth
Application Process
Candidates can attend the walk-in interview directly.
Walk-In Details:
- Date: 25 April 2026
- Time: 9:00 AM to 12:30 PM
Interview Venue:
Cohance Lifesciences Limited
4th Floor, CDC Towers
Nacharam, Hyderabad, Telangana – 500076
Contact Details:
- Phone: 9154672391
Documents Required:
- Updated resume
- Educational certificates (photocopies)
- Last 3 months payslips and CTC structure
Apply early as multiple candidates are expected.
Why Join Cohance Lifesciences?
- Opportunity to work in GMP-compliant formulation plants
- Exposure to OSD manufacturing and QC analytical systems
- Strong career growth in QA, QC, and production domains
- Work with experienced teams in regulated pharma environments
These roles directly contribute to pharmaceutical product development, quality assurance, and global healthcare delivery.
FAQs
What experience is required?
Candidates with 3–6 years of pharma experience can apply.
Are multiple roles available?
Yes, roles are open across Production, QA, QC, Warehouse, and Engineering.
Is formulation experience mandatory?
Yes, candidates should have experience in formulation plants.
What skills are required for QC roles?
Hands-on experience in HPLC and GC analysis.
How to attend the interview?
Visit the venue with required documents on the mentioned date.
Final Thoughts
This Cohance Lifesciences hiring drive is a strong opportunity for experienced pharmaceutical professionals to advance their careers in manufacturing, quality control, and regulatory compliance. With multiple openings and exposure to regulated environments, this is a high-value opportunity.
Attend early to increase your chances of selection.
| Company | Cohance Lifesciences Limited |
|---|---|
| Vacancies | Production Operator, Production Executive, QA Officer, QC Analyst, Warehouse Executive, Engineering Executive |
| Required Education | B.Pharm, M.Pharm, B.Sc, M.Sc, Diploma, ITI |
| Experience | 3 to 6 Years |