Emcure Walk-In QA, Production, Packing, Warehouse & Engineering

- Company Overview
- Job Role & Responsibilities
- Production Department
- Key Skills & Responsibilities
- Responsibilities
- Packing Department
- Operator
- Key Responsibilities
- Officer / Executive
- Responsibilities
- Engineering Department
- Operator
- Areas & Skills
- Officer
- Responsibilities
- Executive / Assistant Manager
- Responsibilities
- Warehouse Department
- Operator
- Responsibilities
- Officer
- Responsibilities
- Technology Transfer Department
- Officer / Executive
- Key Skills
- Responsibilities
- Quality Assurance Department
- Officer / Executive
- Key Responsibilities
- Eligibility / Qualifications
- Production Department
- Packing Department
- Engineering Department
- Warehouse Department
- Technology Transfer & Quality Assurance
- Relevant Courses
- Experience Required
- Preferred Skills
- Why This Opportunity Is Important for Pharma Professionals
- Location & Salary
- Job Location
- Salary
- Application Process
- Walk-In Interview Details
- Interview Venue
- Documents Required
- Contact Details
- Contact Numbers
- Why Candidates Should Apply?
- FAQs
- 1. Which company is conducting the walk-in drive?
- 2. What is the job location?
- 3. Which departments have openings?
- 4. Are freshers eligible for any roles?
- 5. What qualifications are accepted?
- 6. What documents should candidates carry?
- Summary Table
Emcure Pharma Walk-In | QA Production Jobs
Emcure Pharmaceuticals hiring for QA, Production, Packing, Warehouse & Engineering roles at Sanand Injectable Plant. Fresher & experienced eligible.
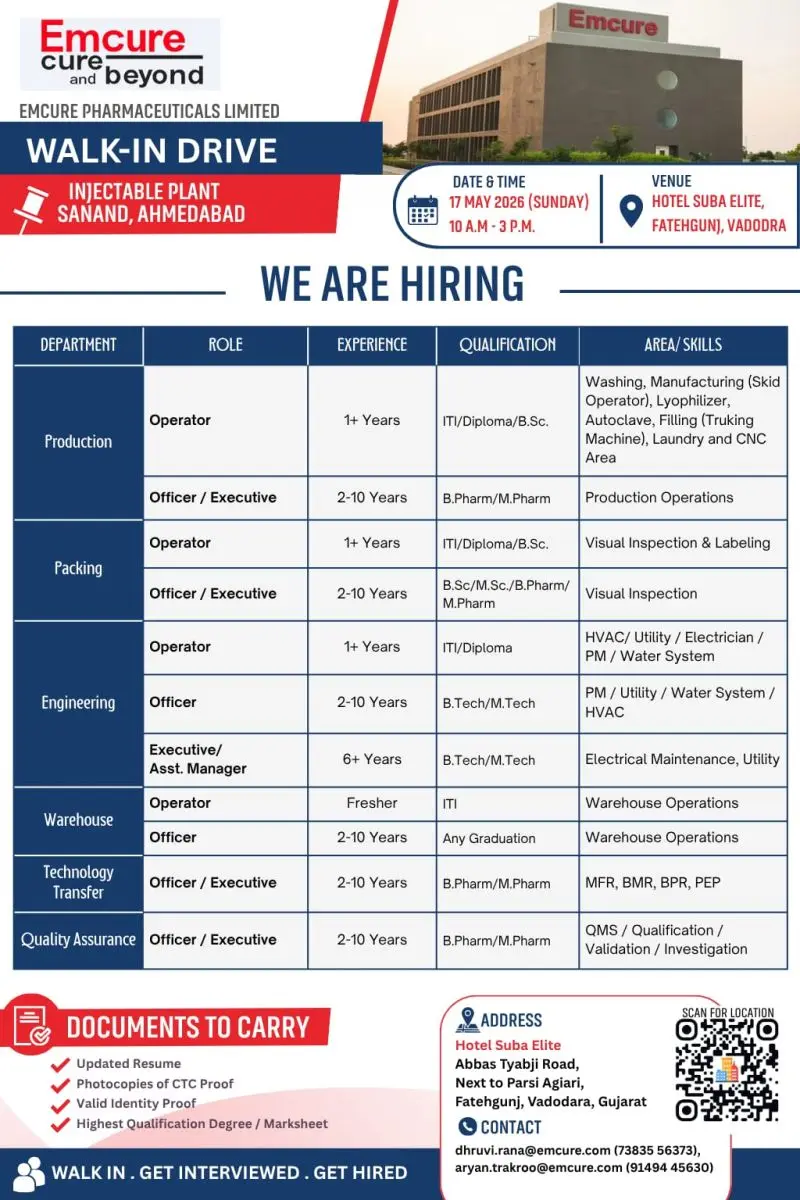
Emcure Pharmaceuticals Limited has announced a major walk-in hiring drive for its Injectable Plant located at Sanand, Ahmedabad. The company is inviting applications from experienced pharmaceutical professionals and fresh candidates for multiple departments including Production, Packing, Engineering, Warehouse, Technology Transfer, and Quality Assurance.
This pharmaceutical walk-in interview is an excellent opportunity for candidates searching for injectable pharma jobs, QA jobs in pharma, production operator vacancies, warehouse pharma jobs, engineering jobs in pharmaceutical companies, and B.Pharm M.Pharm walk-in interviews in Gujarat.
Candidates with experience in injectable manufacturing operations, quality systems, technology transfer, HVAC utilities, warehouse operations, and pharmaceutical production processes are encouraged to attend the interview drive.
The hiring process is scheduled in Vadodara for positions based at the company’s Sanand Injectable Plant near Ahmedabad.
Company Overview
Emcure Pharmaceuticals Limited is one of India’s leading pharmaceutical companies with a strong presence across domestic and international pharmaceutical markets. The company is known for its expertise in formulations, injectables, biologics, healthcare innovation, and regulated pharmaceutical manufacturing.
Emcure operates advanced pharmaceutical manufacturing facilities compliant with international regulatory standards and quality systems. The company focuses on innovation-driven manufacturing, high-quality healthcare products, and strong pharmaceutical compliance practices.
The injectable manufacturing segment requires highly skilled professionals capable of handling aseptic processing, quality management systems, engineering utilities, validation activities, visual inspection systems, and pharmaceutical documentation.
Professionals joining Emcure Pharmaceuticals gain exposure to regulated pharmaceutical manufacturing environments, GMP-compliant operations, advanced injectable technologies, and cross-functional pharma operations.
Job Role & Responsibilities
Production Department
Operator
Candidates selected for Production Operator roles will support injectable manufacturing activities in regulated production environments.
Key Skills & Responsibilities
- Handling washing and manufacturing operations
- Operating skid systems and injectable equipment
- Managing lyophilizer operations
- Handling autoclave systems
- Supporting filling operations including truking machine processes
- Working in laundry and CNC areas
- Following GMP and pharmaceutical safety guidelines
- Maintaining batch documentation and production records
Officer / Executive
Production Officers and Executives will handle injectable production operations, manpower coordination, documentation activities, and manufacturing process compliance.
Responsibilities
- Supervise injectable manufacturing operations
- Ensure compliance with cGMP guidelines
- Coordinate production activities with QA and Engineering teams
- Maintain production documentation and batch records
- Ensure smooth execution of manufacturing schedules
Packing Department
Operator
Packing Operators will work in visual inspection and labeling sections.
Key Responsibilities
- Perform visual inspection of injectable products
- Handle labeling and packaging operations
- Ensure packing quality standards
- Maintain documentation related to packaging activities
- Support production and QA coordination
Officer / Executive
Packing Officers and Executives will supervise visual inspection processes and injectable packaging operations.
Responsibilities
- Monitor visual inspection activities
- Ensure packaging compliance with GMP standards
- Handle packaging documentation and reconciliation
- Coordinate packing line operations and manpower activities
Engineering Department
Operator
Engineering Operators will support utility and maintenance activities in the pharmaceutical facility.
Areas & Skills
- HVAC systems
- Utility maintenance
- Electrical systems
- Preventive maintenance
- Water systems
Officer
Engineering Officers will manage pharmaceutical utility systems and maintenance operations.
Responsibilities
- Handle preventive maintenance operations
- Monitor HVAC and utility systems
- Ensure smooth functioning of pharmaceutical utilities
- Support qualification and maintenance documentation
Executive / Assistant Manager
Senior engineering professionals will manage electrical maintenance and utility systems within the injectable manufacturing plant.
Responsibilities
- Supervise utility operations
- Handle electrical maintenance planning
- Ensure regulatory and GMP compliance
- Support engineering troubleshooting and process improvements
Warehouse Department
Operator
Warehouse Operators will support pharmaceutical warehouse and material handling operations.
Responsibilities
- Handle warehouse documentation
- Manage material movement activities
- Support inventory handling and storage
- Follow warehouse SOPs and compliance standards
Officer
Warehouse Officers will supervise warehouse operations and inventory management systems.
Responsibilities
- Monitor pharmaceutical inventory systems
- Coordinate warehouse logistics and dispatch operations
- Ensure compliance with storage guidelines
- Maintain warehouse documentation records
Technology Transfer Department
Officer / Executive
Technology Transfer professionals will support manufacturing transfer and pharmaceutical documentation activities.
Key Skills
- MFR preparation
- BMR handling
- BPR documentation
- PEP documentation
Responsibilities
- Support technology transfer activities
- Prepare manufacturing documentation
- Coordinate with cross-functional departments
- Ensure documentation compliance and batch readiness
Quality Assurance Department
Officer / Executive
QA professionals will manage pharmaceutical quality systems and validation activities.
Key Responsibilities
- Handle QMS activities
- Manage qualification and validation processes
- Support investigation and compliance activities
- Review pharmaceutical documentation
- Participate in GMP audits and regulatory readiness activities
Candidates with injectable manufacturing exposure, pharmaceutical compliance knowledge, and quality management expertise may receive preference.
Eligibility / Qualifications
Production Department
- ITI
- Diploma
- B.Sc
- B.Pharm
- M.Pharm
Packing Department
- ITI
- Diploma
- B.Sc
- M.Sc
- B.Pharm
- M.Pharm
Engineering Department
- ITI
- Diploma
- B.Tech
- M.Tech
Warehouse Department
- Any Graduation
Technology Transfer & Quality Assurance
- B.Pharm
- M.Pharm
Relevant Courses
B.Pharm, M.Pharm, Diploma in Pharmacy, B.Sc Chemistry, M.Sc Chemistry, Mechanical Engineering, Electrical Engineering, Pharmaceutical Technology, Industrial Pharmacy, Pharmaceutical Quality Assurance, Production Engineering, HVAC Maintenance, Utility Engineering
Experience Required
- Fresher to 10 Years depending on role
Preferred Skills
- Injectable manufacturing exposure
- GMP documentation knowledge
- Validation and qualification understanding
- Warehouse inventory handling
- HVAC and utility maintenance
- Pharmaceutical compliance systems
- Visual inspection and packaging operations
- Technology transfer documentation
- QMS handling experience
Why This Opportunity Is Important for Pharma Professionals
Injectable manufacturing is one of the most regulated and technically advanced segments in the pharmaceutical industry. Professionals working in injectable facilities gain strong exposure to:
- Aseptic manufacturing systems
- Pharmaceutical quality compliance
- Validation and qualification activities
- Utility and engineering operations
- Injectable packaging systems
- Technology transfer processes
- Regulatory audit preparedness
- Pharmaceutical warehouse management
Emcure Pharmaceuticals offers candidates the opportunity to work in advanced pharmaceutical manufacturing environments with exposure to global pharmaceutical standards.
Location & Salary
Job Location
Sanand, Ahmedabad
Salary
Salary will be offered according to candidate experience, technical expertise, and company standards.
Application Process
Interested candidates can directly attend the walk-in interview with required documents.
Walk-In Interview Details
- Date: 17 May 2026 (Sunday)
- Time: 10:00 AM to 3:00 PM
Interview Venue
Hotel Suba Elite, Fatehgunj, Vadodara, Gujarat
Documents Required
Candidates should carry:
- Updated Resume
- Photocopies of CTC Proof
- Valid Identity Proof
- Highest Qualification Degree / Marksheet
Contact Details
Contact Numbers
- 73835 56373
- 91494 45630
Why Candidates Should Apply?
- Opportunity to work at a leading injectable pharmaceutical plant
- Openings across Production, QA, Engineering, Warehouse, and Technology Transfer
- Strong career growth opportunities in regulated pharma manufacturing
- Exposure to injectable pharmaceutical operations
- Opportunities for both freshers and experienced professionals
- Work in GMP-compliant pharmaceutical environments
- Gain experience in regulated manufacturing systems and documentation
FAQs
1. Which company is conducting the walk-in drive?
Emcure Pharmaceuticals Limited is conducting the walk-in drive for its Injectable Plant.
2. What is the job location?
Selected candidates will work at the Sanand Injectable Plant near Ahmedabad.
3. Which departments have openings?
Openings are available in Production, Packing, Engineering, Warehouse, Technology Transfer, and Quality Assurance.
4. Are freshers eligible for any roles?
Yes. Freshers are eligible for Warehouse Operator positions.
5. What qualifications are accepted?
ITI, Diploma, B.Sc, M.Sc, B.Pharm, M.Pharm, B.Tech, and M.Tech candidates can apply depending on the role.
6. What documents should candidates carry?
Applicants should carry updated resumes, qualification certificates, ID proof, and CTC documents.
Summary Table
| Company | Emcure Pharmaceuticals Limited |
| Department Vacancies | Production, Packing, Engineering, Warehouse, QA, Technology Transfer |
| Qualification | ITI, Diploma, B.Sc, M.Sc, B.Pharm, M.Pharm, B.Tech, M.Tech |
| Experience | Fresher to 10 Years |
| Location | Sanand, Ahmedabad |
You must sign in to apply for this position.


