Honour walk-in PR&D, QC, QA & Production

- Company Overview
- Job Role & Responsibilities
- PR&D – Process Research & Development
- Quality Control (QC)
- Quality Assurance (QA)
- Production – Officer to Sr. Executive
- Production – Junior Officer (Freshers)
- QC/QA – Fresher Officer Roles
- Eligibility / Qualifications
- Educational Qualification
- Relevant Courses
- Experience Required
- Preferred Technical Skills
- Why API Manufacturing Careers Are in High Demand
- Work Environment & Career Growth
- Location & Salary
- Job Location
- Work Location
- Approximate Salary
- Application Process
- Walk-In Interview Dates
- Interview Timing
- Walk-In Venue
- Contact Details
- Official Website
- Documents to Carry
- Important Note
- FAQs
- 1. Which company is conducting this pharma walk-in interview?
- 2. Which departments are hiring?
- 3. Are freshers eligible for Honour Synthesis jobs?
- 4. What qualifications are eligible for these pharma jobs?
- 5. What is the job location?
- 6. What skills are preferred for experienced candidates?
- Summary Table
- Walkins
- 8-3-166/7/5, Vikaspuri, Beside Bata Showroom, Erragadda, Hyderabad, Telangana – 500038
- May 17, 2026
Honour Synthesis QC QA Jobs | MSc Chemistry
Honour Synthesis hiring freshers & experienced candidates for PR&D, QC, QA & Production roles in Hyderabad/Sangareddy.
Honour Synthesis Pvt. Ltd. has announced a major walk-in interview drive for experienced and fresher pharmaceutical professionals across PR&D, Production, Quality Control, and Quality Assurance departments. Candidates looking for pharma jobs in Hyderabad, API manufacturing careers, GMP pharmaceutical jobs, QC analyst jobs, production executive roles, and process development careers can explore this hiring opportunity.
The recruitment drive will be conducted for the company’s Sangareddy manufacturing units, offering opportunities for both freshers and experienced professionals with Chemistry backgrounds. Honour Synthesis is actively hiring candidates with expertise in GMP-regulated pharmaceutical manufacturing environments, quality systems, process development, analytical testing, and batch manufacturing operations.
This latest pharma walk-in drive is expected to attract candidates searching for high-growth pharmaceutical careers in API manufacturing, quality assurance, regulatory compliance, analytical development, and production operations.
Company Overview
Honour Synthesis Pvt. Ltd. is a leading pharmaceutical CDMO company engaged in API manufacturing, pharmaceutical process development, quality systems management, and regulated pharmaceutical operations.
The company operates advanced manufacturing facilities focused on maintaining strong compliance with GMP regulations, pharmaceutical quality systems, regulatory documentation standards, and global manufacturing practices.
Honour Synthesis has built a reputation for handling complex pharmaceutical manufacturing operations while maintaining strict compliance with:
- GMP guidelines
- Regulatory quality systems
- Process validation standards
- Pharmaceutical documentation practices
- Analytical quality control systems
- Technology transfer protocols
- Risk assessment frameworks
- Quality management systems
The company offers pharmaceutical professionals exposure to:
- API manufacturing operations
- Process research and development
- Analytical quality control
- Technology transfer activities
- Validation and qualification systems
- QMS investigations
- Vendor management systems
- Regulatory compliance documentation
With the increasing demand for regulated pharmaceutical manufacturing and API exports, experienced professionals in QC, QA, PR&D, and Production continue to remain highly valuable across the Indian pharmaceutical industry.
Job Role & Responsibilities
Honour Synthesis is hiring candidates across multiple departments including PR&D, Quality Control, Quality Assurance, and Production.
Selected candidates will work in regulated GMP environments involving pharmaceutical manufacturing, process development, quality systems management, analytical testing, and production operations.
PR&D – Process Research & Development
Candidates selected for PR&D roles will support pharmaceutical process development activities and manufacturing process optimization.
Key Responsibilities
- Handling process development activities
- Executing experiments in GMP environments
- Supporting scale-up activities
- Handling laboratory glassware and chemical processes
- Process optimization and troubleshooting
- Supporting technology transfer documentation
- Assisting process validation activities
- Maintaining process development records
- Following safety and GMP procedures
Candidates should possess strong chemistry knowledge and understanding of pharmaceutical process development operations.
Quality Control (QC)
Quality Control professionals will manage analytical testing activities and laboratory quality systems.
Key Responsibilities
- Operating HPLC and GC instruments
- Handling AMV and method transfer activities
- Reviewing analytical documents and reports
- Managing electronic quality systems like LIMS, DMS, QMS, and WMPS
- Supporting laboratory investigations
- Instrument calibration and maintenance coordination
- Regulatory documentation support
- Stability and analytical testing activities
- Maintaining GMP laboratory compliance
Candidates with strong analytical instrumentation exposure and regulatory documentation experience may receive preference.
Quality Assurance (QA)
QA professionals will manage compliance systems, validation activities, and pharmaceutical quality operations.
Key Responsibilities
- Handling technology transfer activities
- Managing vendor qualification systems
- Supporting process and cleaning validation
- Qualification and compliance activities
- QMS investigations and deviation handling
- Documentation review and approval
- Risk assessment management
- Regulatory audit support
- Ensuring GMP compliance across operations
Candidates with pharmaceutical quality systems exposure and regulatory compliance knowledge are encouraged to attend the walk-in interview.
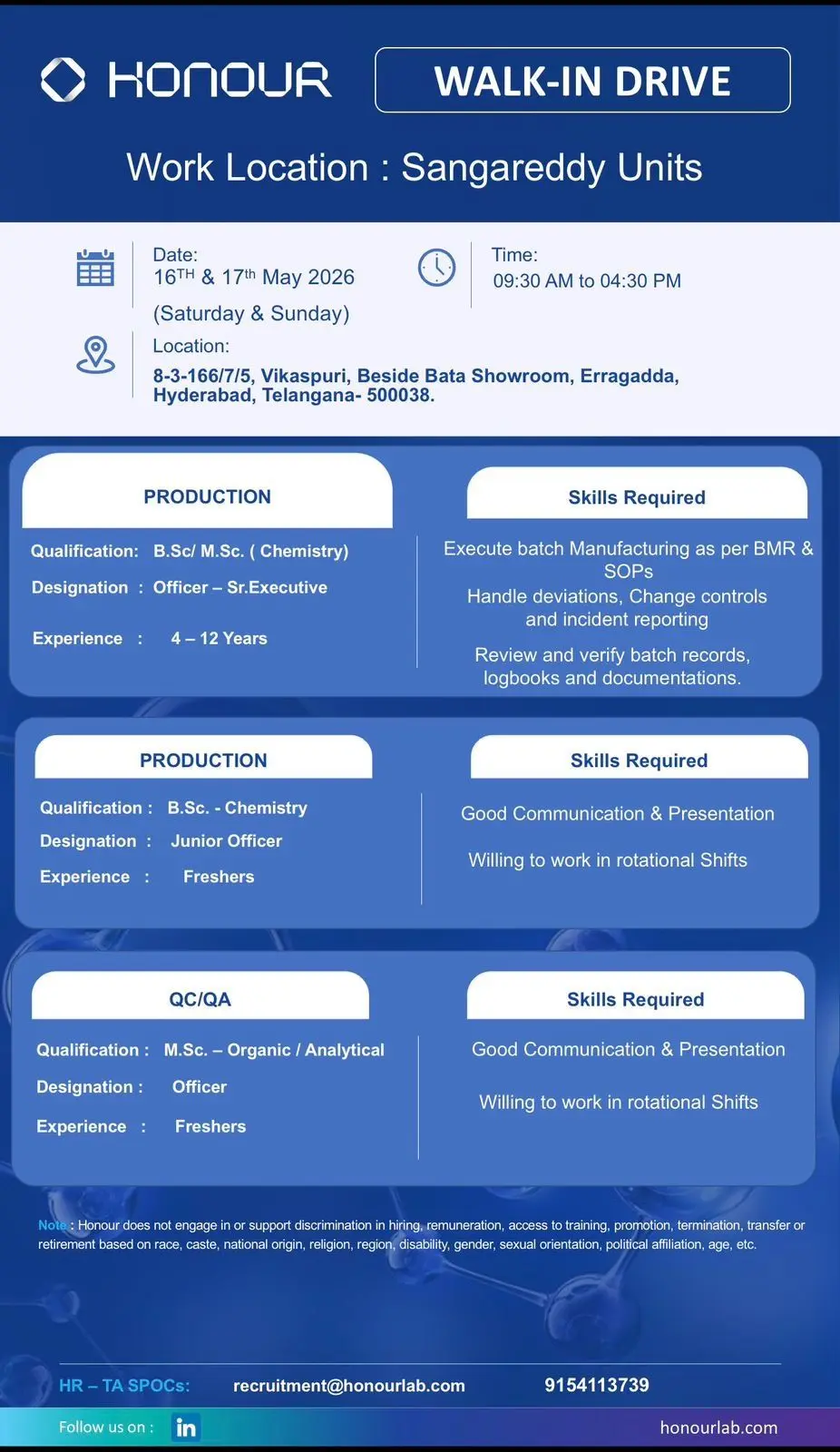
Production – Officer to Sr. Executive
Production professionals will support API manufacturing and batch execution activities.
Key Responsibilities
- Executing batch manufacturing activities as per BMR and SOPs
- Handling change controls and deviations
- Incident reporting and investigation support
- Batch record review and verification
- Production documentation maintenance
- Logbook review and GMP documentation
- Supporting shift manufacturing activities
- Following regulatory production procedures
Candidates with API production experience and GMP exposure may receive preference.
Production – Junior Officer (Freshers)
Freshers selected for Production roles may receive exposure to pharmaceutical manufacturing operations and production systems.
Key Responsibilities
- Supporting production operations
- Learning GMP documentation practices
- Following SOP-based manufacturing activities
- Coordinating shift production tasks
- Supporting batch execution activities
- Maintaining shop floor discipline
Freshers should possess good communication skills and willingness to work in rotational shifts.
QC/QA – Fresher Officer Roles
Freshers joining QC or QA departments may support laboratory and quality assurance operations under experienced professionals.
Key Responsibilities
- Supporting analytical documentation
- Assisting laboratory activities
- Supporting quality systems management
- Learning GMP and compliance procedures
- Assisting documentation review processes
- Supporting audit preparation activities
Eligibility / Qualifications
Candidates applying for these pharmaceutical jobs should satisfy the eligibility criteria mentioned below.
Educational Qualification
PR&D
- B.Sc Chemistry
- M.Sc Chemistry
Quality Control
- B.Sc Chemistry
- M.Sc Chemistry
Quality Assurance
- B.Sc Chemistry
- M.Sc Chemistry
Production
- B.Sc Chemistry
- M.Sc Chemistry
Fresher QC/QA Roles
- M.Sc Organic Chemistry
- M.Sc Analytical Chemistry
Relevant Courses
Organic Chemistry, Analytical Chemistry, Pharmaceutical Chemistry, Industrial Chemistry, Process Chemistry, Pharmaceutical Analysis, API Manufacturing Technology, Quality Assurance, Pharmaceutical Quality Systems, GMP Compliance, Industrial Pharmacy
Experience Required
PR&D
- 4 to 12 Years
Quality Control
- 6 to 12 Years
Quality Assurance
- 6 to 12 Years
Production Experienced Roles
- 4 to 12 Years
Production Fresher Roles
- Freshers Eligible
QC/QA Fresher Roles
- Freshers Eligible
Preferred Technical Skills
Candidates with the following technical skills may receive preference:
- API manufacturing
- GMP documentation
- HPLC operations
- GC analysis
- Method validation
- Method transfer
- QMS handling
- Technology transfer
- Process validation
- Cleaning validation
- Batch manufacturing
- SOP compliance
- Deviation handling
- Change control systems
- Regulatory compliance
- Analytical instrumentation
Why API Manufacturing Careers Are in High Demand
The pharmaceutical API manufacturing sector continues to expand due to increasing global demand for regulated pharmaceutical products and active pharmaceutical ingredients.
Professionals with expertise in API production, quality systems, analytical chemistry, and process development are highly sought after by:
- Pharmaceutical CDMO companies
- API manufacturers
- Regulated export facilities
- USFDA-compliant plants
- Global pharmaceutical manufacturing companies
- Research and development organizations
Candidates working in GMP-regulated pharmaceutical environments gain long-term career advantages in:
- Production management
- Quality systems leadership
- Regulatory affairs
- Process development
- Technology transfer
- Analytical research
- Validation and compliance
Work Environment & Career Growth
Honour Synthesis offers candidates exposure to modern pharmaceutical manufacturing systems and regulated production environments.
Professionals joining the organization may receive opportunities to work on:
- API process development
- Analytical quality systems
- GMP manufacturing operations
- Regulatory compliance programs
- Technology transfer activities
- Validation systems
- Pharmaceutical quality management
- Risk assessment and investigations
Employees with strong technical expertise and regulatory understanding may grow into senior production, QA, QC, validation, and process development roles.
Location & Salary
Job Location
Hyderabad
Work Location
Sangareddy Units
Approximate Salary
Based on current pharmaceutical industry salary trends in Hyderabad and Sangareddy API manufacturing companies, the approximate salary range may vary between ₹2.8 LPA to ₹12 LPA depending on experience, department, technical skills, and qualification.
Application Process
Eligible candidates can directly attend the walk-in interview at the venue mentioned below.
Walk-In Interview Dates
16th & 17th May 2026
Interview Timing
09:00 AM to 04:30 PM
Walk-In Venue
8-3-166/7/5, Vikaspuri, Beside Bata Showroom, Erragadda, Hyderabad, Telangana – 500038
Contact Details
- Email: recruitment@honourlab.com
- Contact Number: 9154113739
Official Website
Documents to Carry
Candidates should preferably carry:
- Updated resume
- Educational certificates
- Experience certificates
- Government ID proof
- Passport-size photographs
- Salary documents for experienced candidates
Important Note
Honour Synthesis Pvt. Ltd. follows equal employment opportunity practices and does not support discrimination in hiring, promotion, remuneration, training, transfer, or retirement.
The company encourages eligible pharmaceutical professionals and freshers to attend the walk-in drive directly.
FAQs
1. Which company is conducting this pharma walk-in interview?
Honour Synthesis Pvt. Ltd. is conducting the walk-in drive for its Sangareddy units.
2. Which departments are hiring?
The company is hiring for PR&D, Production, Quality Control, and Quality Assurance departments.
3. Are freshers eligible for Honour Synthesis jobs?
Yes. Freshers are eligible for Junior Officer and QC/QA Officer roles.
4. What qualifications are eligible for these pharma jobs?
B.Sc Chemistry, M.Sc Chemistry, M.Sc Organic Chemistry, and M.Sc Analytical Chemistry candidates are eligible.
5. What is the job location?
The work location is Sangareddy, while the walk-in venue is in Hyderabad.
6. What skills are preferred for experienced candidates?
API manufacturing, HPLC, GMP compliance, method validation, QMS handling, process development, and technology transfer experience are preferred.
Summary Table
| Company | Honour Synthesis Pvt. Ltd. |
| Department Vacancies | PR&D, QC, QA, Production |
| Qualification | B.Sc Chemistry, M.Sc Chemistry, M.Sc Organic, M.Sc Analytical |
| Experience | Freshers to 12 Years |
| Location | Hyderabad / Sangareddy |

You must sign in to apply for this position.


