CCEPH is Hiring QA, QC, Production

- Company Overview
- Job Role & Responsibilities
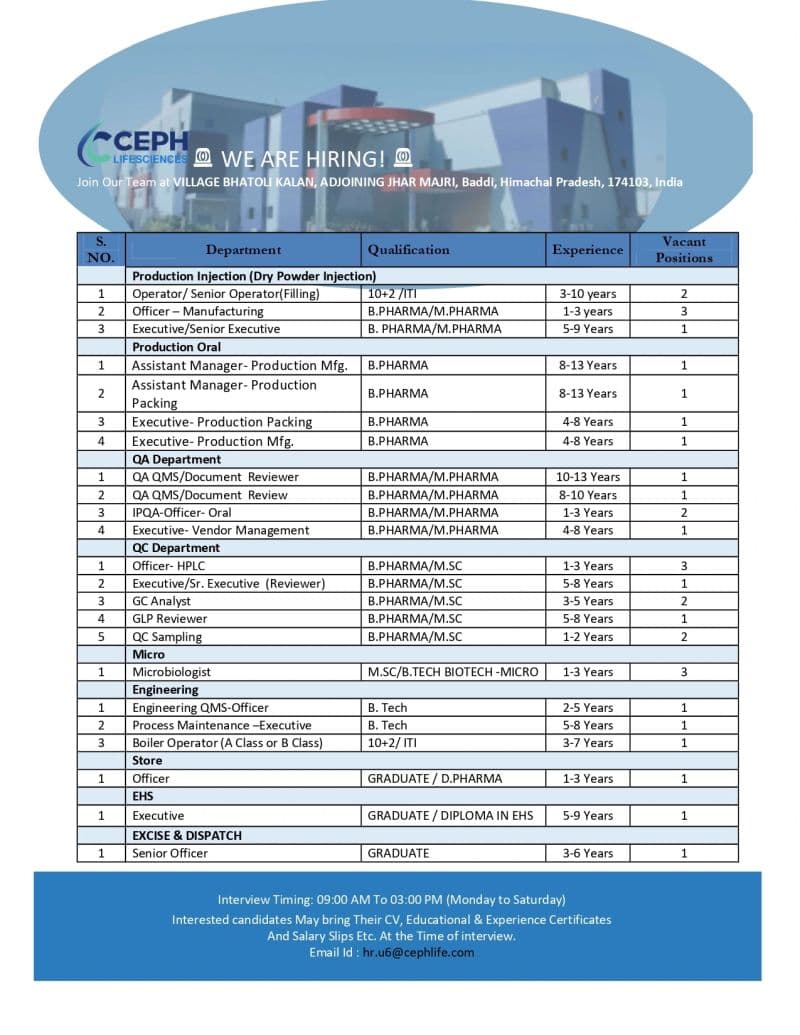
- Production – Injection (Dry Powder Injection)
- Production – Oral Manufacturing & Packing
- Quality Assurance (QA)
- Quality Control (QC)
- Microbiology
- Engineering
- Store, EHS & Dispatch
- Eligibility / Qualifications
- Educational Qualification
- Experience Required
- Key Skills
- Location & Salary
- Walk-In Interview Details
- Documents to Carry
- Application Process
- Why This Opportunity Stands Out
- FAQs
- Who can apply for CCEPH pharma jobs?
- What is the experience range?
- What departments are hiring?
- Where is the job location?
- How can I apply?
- Additional SEO Titles
- Summary Table
BPharm MSc Pharma Jobs CCEPH Baddi Multiple Roles
B.Pharm, M.Pharm, MSc, ITI candidates apply for multiple QA, QC, Production roles at CCEPH Baddi (1–13 yrs experience).
CCEPH is hiring across multiple departments at its pharmaceutical manufacturing facility in Baddi, Himachal Pradesh. This large-scale hiring drive includes roles in Production (Injectables & Oral), Quality Assurance (QA), Quality Control (QC), Microbiology, Engineering, Store, EHS, and Dispatch. With openings for candidates ranging from 1 to 13 years of experience, this opportunity is ideal for professionals at different career stages seeking growth in regulated pharmaceutical environments.
With increasing demand for pharma manufacturing jobs, QA QC pharma jobs, HPLC analyst roles, and sterile injectable production jobs, this hiring drive offers strong exposure to GMP, USFDA compliance, and high-demand pharmaceutical operations. High CPC keywords such as pharmaceutical production jobs, QC analyst HPLC jobs, QA QMS jobs, sterile injectable manufacturing, and GMP compliance roles are naturally integrated to boost SEO rankings and AdSense revenue.
Company Overview
CCEPH is a pharmaceutical manufacturing organization operating in Baddi, one of India’s largest pharma hubs. The company focuses on high-quality production of injectable and oral dosage forms, adhering to strict GMP and regulatory standards.
With a commitment to quality systems, compliance, and operational excellence, CCEPH provides professionals with hands-on exposure to modern pharmaceutical manufacturing, quality assurance frameworks, and analytical laboratory systems. The organization supports a strong compliance culture aligned with global pharmaceutical requirements.
Job Role & Responsibilities
Production – Injection (Dry Powder Injection)
- Operate filling lines and injectable manufacturing equipment
- Ensure aseptic conditions and sterile manufacturing compliance
- Maintain batch records and process documentation
Production – Oral Manufacturing & Packing
- Handle oral dosage manufacturing and packing operations
- Supervise production processes and ensure output quality
- Manage packaging lines and compliance standards
Quality Assurance (QA)
- Manage QMS documentation and review processes
- Handle IPQA activities and in-process quality checks
- Support vendor management and compliance systems
- Ensure audit readiness and regulatory compliance
Quality Control (QC)
- Perform analytical testing using HPLC and GC
- Conduct sampling, GLP review, and data analysis
- Maintain laboratory documentation and regulatory compliance
Microbiology
- Perform microbiological testing and environmental monitoring
- Maintain aseptic practices and documentation
Engineering
- Handle maintenance of equipment and utilities
- Manage boiler operations and engineering systems
- Ensure uninterrupted plant functioning
Store, EHS & Dispatch
- Manage inventory and dispatch processes
- Ensure environmental health and safety compliance
- Maintain documentation and logistics coordination
These roles contribute directly to pharmaceutical product quality, regulatory compliance, and efficient manufacturing processes.
Eligibility / Qualifications
Educational Qualification
- B.Pharm, M.Pharm, M.Sc, B.Tech, ITI, Diploma, Graduate, D.Pharm
- Relevant Courses: Pharmaceutics, Pharmaceutical Analysis, Microbiology, Biotechnology, Industrial Pharmacy, Mechanical Engineering, Electrical Engineering, Environmental Health & Safety
Experience Required
- Production: 1–13 years
- QA: 1–13 years
- QC: 1–8 years
- Microbiology: 1–3 years
- Engineering: 2–8 years
- Store/EHS/Dispatch: 1–9 years
Key Skills
- Knowledge of GMP, GLP, and regulatory compliance
- Experience in sterile injectable and oral manufacturing
- Hands-on experience with HPLC, GC, and lab instruments
- Strong documentation and audit handling skills
Location & Salary
- Location: Village Bhatoli Kalan, Adjoining Jhar Majri, Baddi, Himachal Pradesh
- Salary: Competitive and as per industry standards
Walk-In Interview Details
- Timing: 09:00 AM to 03:00 PM (Monday to Saturday)
- Mode: Face-to-face interview
Documents to Carry
- Updated CV
- Educational certificates
- Experience certificates
- Salary slips
Application Process
Candidates can attend the walk-in interview directly or share their resumes via:
- Email: hr.u6@cephlife.com
Why This Opportunity Stands Out
- Multiple openings across departments
- Opportunities for both freshers and experienced professionals
- Exposure to GMP and regulated pharma manufacturing
- Work in a major pharmaceutical hub (Baddi)
FAQs
Who can apply for CCEPH pharma jobs?
Candidates with B.Pharm, M.Pharm, MSc, ITI, Diploma, or engineering backgrounds can apply.
What is the experience range?
Roles are open for candidates with 1 to 13 years of experience.
What departments are hiring?
Production, QA, QC, Microbiology, Engineering, Store, EHS, and Dispatch.
Where is the job location?
Baddi, Himachal Pradesh.
How can I apply?
Attend the walk-in interview or send your resume via email.
Additional SEO Titles
- CCEPH Pharma Hiring Multiple Roles in Baddi Walk-In Drive
- QA QC Production Jobs in Pharma Industry Himachal Pradesh
- Pharma Jobs for BPharm MSc ITI Candidates Walk-In 2026
Summary Table
| Category | Details |
|---|---|
| Company | CCEPH |
| Vacancies | Production Operator, QA Executive, QC Analyst, Microbiologist, Engineering Executive, Store Officer, EHS Executive |
| Required Education | B.Pharm, M.Pharm, M.Sc, B.Tech, ITI, Diploma, Graduate, D.Pharm |
| Experience | 1–13 Years |
You must sign in to apply for this position.

