Stallion Hiring Sr Manager & QC Roles

- Stallion Laboratories Sr Manager & QC Roles – USFDA OSD
- Company Overview
- Important Note
- Open Positions (Unit‑II, OSD Formulation)
- Job Role & Responsibilities
- Senior Manager — System Governance
- AQA (Analytical Quality Assurance) — AM/DM
- QC‑LIMS Manager / DM
- QMS — AM/DM
- AMT (QC) — Manager
- ADL Head & ADU Manager
- QC Planning / Review / Audit
- Eligibility / Qualifications
- Technical & Soft Skills Required
- Location & Salary
- Application Process
- Why Join Stallion Laboratories?
- FAQs
- Summary Table
Stallion Laboratories Sr Manager & QC Roles – USFDA OSD
Hiring experienced USFDA-exposed OSD professionals (15–20 yrs/10–12 yrs/2–15 yrs) for QA, QC-LIMS, QMS, AQA at Stallion Laboratories Unit‑II.
Stallion Laboratories Pvt. Ltd. is hiring seasoned professionals for its OSD formulation Unit‑II (USFDA & UK‑MHRA approved). These roles demand hands‑on experience in USFDA‑regulated oral solid dosage (OSD) operations, strong data integrity discipline, and proven leadership in quality systems. We prefer local candidates who can join quickly and bring deep regulatory and technical expertise to a plant positioned to enter additional regulated markets.
Company Overview
Stallion Laboratories, incorporated in 1988, is an integrated pharmaceutical formulation manufacturer with a long track record in niche formulation products. Unit‑II is a USFDA and UK‑MHRA approved OSD facility and is progressing toward EU GMP approval. The company operates with a WHO‑GMP mindset across production and analytical labs and maintains GLP‑qualified quality control. Stallion’s focus is uncompromising quality, regulatory readiness, and expansion into regulated export markets.
Important Note
Only candidates with prior working experience in a USFDA‑inspected OSD facility will be considered. Preference will be given to local candidates.
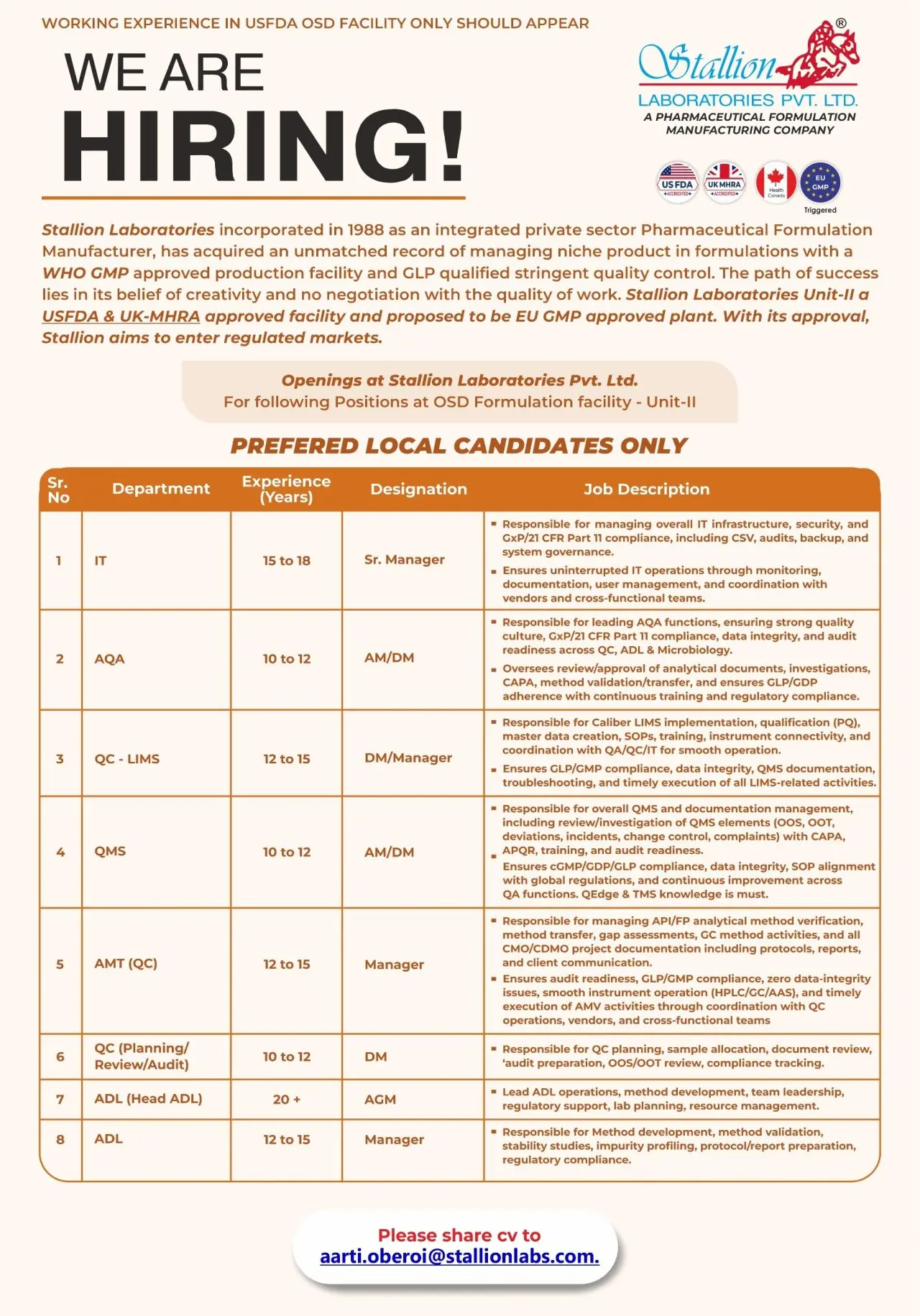
Open Positions (Unit‑II, OSD Formulation)
- System Governance — Sr. Manager (15–18 years)
- AQA (Analytical Quality Assurance) — Assistant Manager/Deputy Manager (10–12 years)
- QC‑LIMS — Deputy Manager/Manager (12–15 years)
- QMS — Assistant Manager/Deputy Manager (10–12 years)
- AMT (QC) — Manager (12–15 years)
- QC Planning & Review / Audit — Officer/Manager (10–12 years)
- ADL (Analytical Development Lab) — Head ADL (20+ years)
- ADU (Analytical Development Unit) — Manager (12–15 years)
Note: Designations and banding above reflect the hiring brief. Exact role title and job grade will be decided during the interview and offer stage.
Job Role & Responsibilities
The roles sit across Quality Assurance, Analytical Development, Laboratory Information Management Systems (LIMS), and Quality Management Systems (QMS). Core responsibilities across the positions include:
Senior Manager — System Governance
- Lead data governance and integrity across QC and ADL in line with 21 CFR Part 11, ALCOA+ principles, and regulatory expectations.
- Define, implement, and monitor electronic records policies, audit trails, and system access controls.
- Drive remediation programs, computerized system validations (CSV), and EW/ELN/LIMS lifecycle management.
- Coordinate cross‑functional compliance activities with QA, QC, IT, and operations.
AQA (Analytical Quality Assurance) — AM/DM
- Oversee analytical document review and approval: protocols, validation reports, stability study reports, and change controls.
- Ensure continuous training programs and audit readiness for analytical teams.
- Lead investigations (OOS/OOT), root cause analysis, CAPA, and supplier quality interactions.
QC‑LIMS Manager / DM
- Manage LIMS administration, user management, electronic signatures, and integration with instruments.
- Ensure data flow integrity from instruments (HPLC, GC, UPLC) to LIMS and ELN; troubleshoot interfacing issues.
- Own periodic data reconciliations, backup strategies, and IT escalation for validated systems.
QMS — AM/DM
- Implement and maintain QMS processes: change control, deviations, CAPA, management review, and internal audits.
- Align SOPs, batch release processes, and documentation practices with MHRA/USFDA expectations.
- Facilitate external audits and regulatory inspections support.
AMT (QC) — Manager
- Lead analytical method development, validation, and transfer for OSD products.
- Supervise routine QC operations: assay, dissolution, content uniformity, impurity profiling, and stability testing.
- Ensure instrument qualification, preventive maintenance, and calibration programs.
ADL Head & ADU Manager
- Drive formulation‑supporting analytical development: assay & impurity methods, stability indicating methods, forced degradation studies, and profiling.
- Coordinate scale‑up analytics during tech transfer and commercialisation.
- Provide regulatory documentation for CMC sections and support responses to queries during audits.
QC Planning / Review / Audit
- Own QC release planning, documentation review, and audit coordination.
- Prepare stability protocols, review stability data, and compile regulatory stability summaries.
- Lead facility audit readiness and manage third‑party audit responses.
Eligibility / Qualifications
- Mandatory: Demonstrable experience working in a USFDA‑inspected OSD facility (oral solid dosage). Applications without this experience will not be considered.
- Experience: 10–20+ years for senior and manager positions; 10–12 years for AM/DM bands; 12–15 years for QC/LIMS and AMT manager roles; 2–7 years not listed here — roles are senior‑focused.
- Education: B.Pharm, M.Pharm, M.Sc (Pharmaceutical Chemistry, Analytical Chemistry), or equivalent. Advanced degrees or specialised certifications in pharmaceutical quality, regulatory affairs, or analytical chemistry are an advantage.
Relevant courses (comma‑separated): B.Pharm, M.Pharm, M.Sc Pharmaceutical Chemistry, M.Sc Analytical Chemistry, M.Sc Organic Chemistry, Diploma in Regulatory Affairs, PG Diploma in Quality Assurance, Certificate in GLP/GMP Compliance, Certification in LIMS/CSV.
Technical & Soft Skills Required
- Strong hands‑on knowledge of HPLC/UPLC, GC, dissolution apparatus, UV, Karl Fischer, and stability chambers.
- Proven track record in method validation, method transfer, and troubleshooting analytical methods.
- Deep understanding of 21 CFR Part 11, EU Annex 11, ALCOA+, data integrity frameworks, and computerized system validation (CSV).
- Experience handling regulatory inspections (USFDA, UK‑MHRA, WHO) and preparing regulatory submissions/support documents.
- Leadership skills, cross‑functional coordination, and the ability to manage high‑pressure inspection scenarios.
Location & Salary
- Location: Stallion Laboratories Unit‑II (OSD formulation facility). Preference for local candidates only.
- Salary: Competitive and commensurate with experience. Exact compensation will be discussed during the offer stage.
Application Process
Please share your CV at: aarti.oberoi@stallionlabs.com
Include the following in your email subject line: Stallion Unit‑II — —
Shortlisted candidates will be contacted for a technical interview. Please ensure your CV clearly states previous USFDA inspection exposure, specific OSD responsibilities, and a summary of regulatory interactions.
Why Join Stallion Laboratories?
- Work at a regulated, inspection‑ready OSD facility with USFDA and UK‑MHRA approvals.
- Be part of the team preparing the plant for EU GMP entry and expansion into regulated markets.
- Influence quality systems and data governance programs that underpin patient safety and product reliability.
- Opportunity to lead cross‑functional projects in analytical development, LIMS transformation, and CSV initiatives.
FAQs
Q: Is USFDA experience mandatory?
A: Yes. Only candidates with prior working experience in a USFDA‑inspected OSD facility will be considered.
Q: Are local candidates preferred?
A: Yes. Preference will be given to local candidates who can join promptly.
Q: How to apply?
A: Email your CV to aarti.oberoi@stallionlabs.com with the subject: “Stallion Unit‑II — — .”
Q: Will remote or contract candidates be considered?
A: These are full‑time, on‑site roles tied to Unit‑II operations. Remote or contract candidates will not be considered.
Q: What documents should I bring for the interview?
A: Bring original certificates, employment proofs showing USFDA OSD facility experience, and any audit/inspection exposure documentation.
Summary Table
| Category | Details |
|---|---|
| Company | Stallion Laboratories Pvt. Ltd. |
| Vacancies | System Governance (Sr. Manager), AQA (AM/DM), QC‑LIMS (DM/Manager), QMS (AM/DM), AMT (QC Manager), QC Planning/Review (Manager), ADL Head, ADU Manager |
| Required Education | B.Pharm, M.Pharm, M.Sc Pharmaceutical Chemistry, M.Sc Analytical Chemistry, M.Sc Organic Chemistry, Diploma/PG in Regulatory Affairs or QA |
| Experience | 10–20+ years depending on role; Mandatory: Experience in a USFDA‑inspected OSD facility |
You must sign in to apply for this position.


