Pharma Hiring Engineering Production QA

- Company Overview
- Job Role & Responsibilities
- Engineering Department
- Production Department (Injectable)
- Formulation & Development (F&D)
- Quality Assurance (QA – IPQA)
- Microbiology Department
- EHS Department
- Eligibility / Qualifications
- Required Education
- Relevant Courses
- Experience
- Skills Required
- Location & Salary
- Job Location
- Salary
- Application Process
- Why You Should Apply
- FAQs
- Are freshers eligible for this hiring?
- What qualifications are required?
- What departments are hiring?
- Is sterile experience required?
- What is the experience range?
- Summary Table
Pharma Company Hiring Multiple Roles Freshers & Experienced Engineering Production QA Jobs Apply Now
A leading pharmaceutical company is hiring for multiple vacancies across Engineering, Production, Quality Assurance (QA), Microbiology, Formulation & Development (F&D), and EHS departments. This recruitment drive includes fresher and experienced roles, offering opportunities for candidates with ITI, Diploma, B.Pharm, M.Pharm, B.Sc, and M.Sc qualifications.
With more than 15+ openings across departments including HVAC Officer, Electrician, Production Officer, QA IPQA Officer, and Microbiology roles, this is a strong opportunity for candidates looking to build or advance their careers in pharmaceutical manufacturing, sterile injectables, and formulation development.
Company Overview
The hiring organization is a pharmaceutical manufacturing company operating in regulated environments with a focus on sterile injectable formulations and oral solid dosage (OSD) development. The company follows strict GMP guidelines and supports production, quality control, and engineering operations aligned with global regulatory standards.
Working in such an environment provides hands-on exposure to pharmaceutical production, utility systems, quality assurance practices, and compliance-driven operations essential for healthcare product safety.
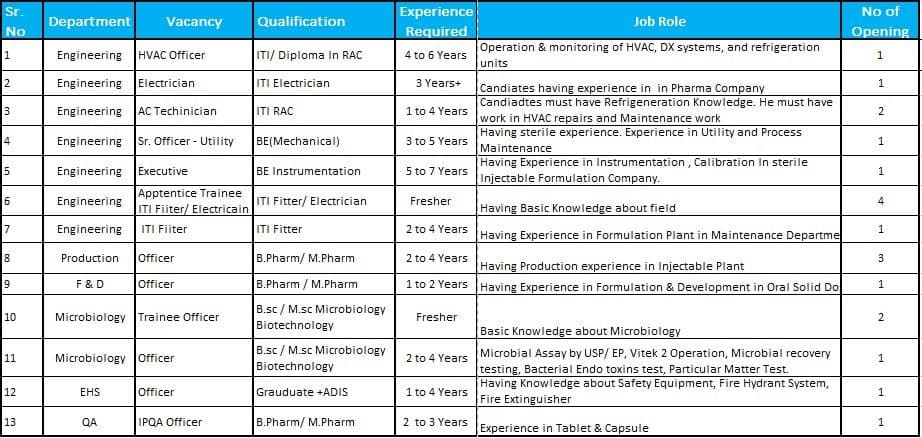
Job Role & Responsibilities
Engineering Department
- Operate and monitor HVAC, DX systems, and refrigeration units
- Perform maintenance of utility systems in sterile manufacturing facilities
- Handle electrical systems, calibration, and instrumentation activities
- Conduct preventive and corrective maintenance of equipment
- Ensure compliance with GMP and safety standards
Production Department (Injectable)
- Handle manufacturing activities in sterile injectable plants
- Maintain batch manufacturing records and production documentation
- Ensure compliance with SOPs and GMP guidelines
- Monitor process parameters and production efficiency
Formulation & Development (F&D)
- Support formulation development of oral solid dosage forms
- Conduct trials, documentation, and product development activities
- Work with R&D and production teams for product scale-up
Quality Assurance (QA – IPQA)
- Perform in-process quality assurance checks
- Ensure compliance with GMP and regulatory standards
- Monitor production processes for quality consistency
Microbiology Department
- Perform microbial testing including BET, MLT, and sterility testing
- Conduct microbial assay as per USP/EP guidelines
- Operate instruments like Vitek 2
- Perform environmental monitoring and microbial recovery testing
EHS Department
- Ensure workplace safety and compliance with safety regulations
- Manage fire safety systems, hydrants, and extinguishers
- Implement safety protocols and risk assessments
Eligibility / Qualifications
Required Education
ITI (Fitter, Electrician, RAC), Diploma (Engineering), B.Pharm, M.Pharm, B.Sc, M.Sc, BE (Mechanical/Instrumentation), Graduate + ADIS
Relevant Courses
Pharmaceutical Engineering, Industrial Pharmacy, Microbiology, Biotechnology, Mechanical Engineering, Instrumentation, Safety Management, HVAC & Refrigeration
Experience
- Freshers: Apprentice Trainee (ITI), Microbiology Trainee
- Engineering Roles: 1 to 7 years
- Production & QA: 2 to 4 years
- Microbiology Officer: 2 to 4 years
- EHS Officer: 1 to 4 years
Skills Required
- Knowledge of GMP, GLP, and pharmaceutical compliance
- Experience in sterile injectable or formulation plants
- Understanding of HVAC, utilities, and maintenance systems
- Analytical and microbiological testing knowledge
- Strong documentation and process control skills
Location & Salary
Job Location
- Pharmaceutical Manufacturing Facility (Location not specified)
Salary
- Competitive salary based on experience and role
Application Process
Interested candidates should apply by sharing their updated resume through the official hiring contact provided by the company (details to be confirmed from HR communication).
Candidates are advised to mention the role applied for and ensure relevant experience details are included in the CV.
Why You Should Apply
- Multiple openings across pharma departments
- Opportunities for both freshers and experienced candidates
- Exposure to sterile injectable and formulation manufacturing
- Career growth in engineering, QA, QC, and production roles
FAQs
Are freshers eligible for this hiring?
Yes, freshers can apply for trainee roles in ITI and Microbiology departments.
What qualifications are required?
ITI, Diploma, B.Pharm, M.Pharm, B.Sc, M.Sc, and Engineering degrees.
What departments are hiring?
Engineering, Production, QA, Microbiology, F&D, and EHS.
Is sterile experience required?
Yes, for many roles, especially production and engineering, sterile injectable experience is preferred.
What is the experience range?
From freshers to candidates with up to 7 years of experience depending on the role.
Summary Table
| Company | Pharmaceutical Manufacturing Company |
|---|---|
| Vacancies | Engineering, Production Officer, QA IPQA Officer, Microbiology Officer |
| Required Education | ITI, Diploma, B.Pharm, M.Pharm, B.Sc, M.Sc, BE |
| Experience | Freshers to 7 years |
You must sign in to apply for this position.

