USV Walk-In Operations, QC, Engineering & Sterile Manufacturing

- Company Overview
- Job Role & Responsibilities
- Operations Department
- Engineering Department – Sterile / Injectables
- QMS Department
- Engineering Department – OSD & Utilities
- Sterile Manufacturing Department
- Quality Control Department
- Finished Goods / Stability Department
- Raw Material Quality Control
- Eligibility / Qualifications
- Required Educational Qualification
- Relevant Courses
- Experience Requirement
- Preferred Skills
- Location & Salary
- Job Location
- Salary
- Application Process
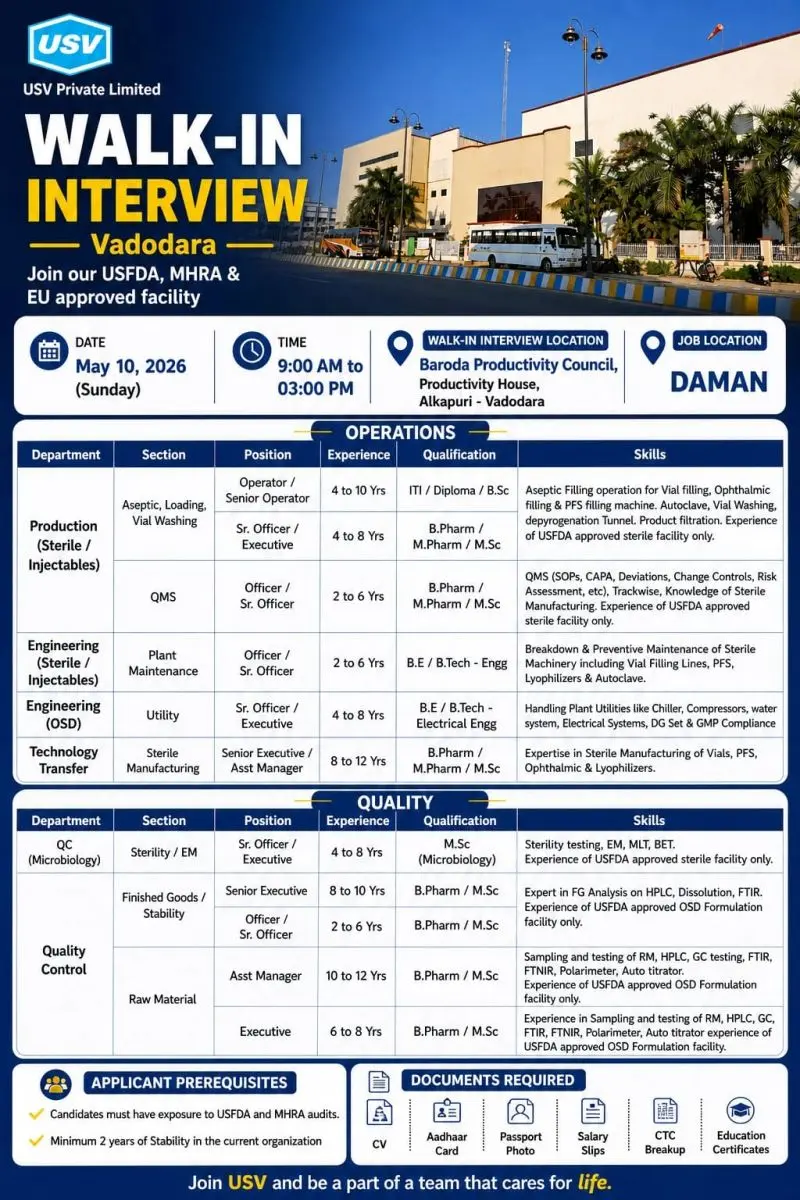
- Walk-In Interview Details
- Walk-In Interview Venue
- Documents Required
- Applicant Prerequisites
- FAQs
- 1. What is the interview date for USV walk-in recruitment?
- 2. What qualifications are required for USV pharma jobs?
- 3. What is the required experience for these vacancies?
- 4. Which departments are hiring at USV?
- 5. What is the job location?
- 6. Is experience in USFDA-approved facilities mandatory?
- Summary Table
USV Walk-In Drive | Pharma Jobs | Daman
USV hiring for Operations, QC, Engineering & Sterile Manufacturing. B.Pharm, M.Pharm, M.Sc eligible in Daman.
USV Private Limited has announced a major walk-in interview drive for experienced pharmaceutical professionals at its USFDA, MHRA, and EU-approved manufacturing facility. The recruitment drive offers opportunities across Operations, Engineering, Quality Control, QMS, Sterile Manufacturing, Technology Transfer, Utilities, and Microbiology departments for candidates with pharmaceutical manufacturing and sterile injectable experience.
Professionals searching for pharma walk-in interviews, sterile injectable jobs, USFDA plant vacancies, quality control careers, engineering jobs in pharmaceutical companies, production officer openings, microbiology jobs, and regulatory manufacturing opportunities can attend this latest hiring drive organized by USV.
The company is looking for skilled professionals with experience in regulated pharmaceutical environments, sterile manufacturing operations, injectables production, OSD formulation, engineering maintenance, and quality systems. Candidates with exposure to USFDA and MHRA audit environments will receive preference during the selection process.
Company Overview
USV Private Limited is one of India’s leading pharmaceutical companies known for manufacturing high-quality formulations and healthcare products for domestic and international markets. The company operates globally approved pharmaceutical manufacturing facilities with a strong focus on quality compliance, regulatory standards, innovation, and advanced healthcare manufacturing.
USV facilities comply with international pharmaceutical standards including USFDA, MHRA, and EU regulations. The organization continues to expand its pharmaceutical manufacturing operations and recruit skilled professionals in sterile injectables, quality assurance, microbiology, engineering, and production management.
The company offers strong career growth opportunities for professionals involved in pharmaceutical manufacturing, regulatory compliance, sterile operations, quality systems, and healthcare production technologies.
Job Role & Responsibilities
Operations Department
Production – Sterile / Injectables
Available Positions
- Operator
- Senior Operator
- Senior Officer
- Executive
Experience Required
- 4 to 10 Years
Key Skills
- Aseptic operations
- Loading activities
- Vial washing
- Aseptic filling operations
- Ophthalmic filling
- PFS filling machine handling
- Autoclave operations
- Depyrogenation tunnel operations
- Product filtration
Candidates must have experience in USFDA-approved sterile manufacturing facilities.
Engineering Department – Sterile / Injectables
Plant Maintenance
Available Positions
- Officer
- Senior Officer
Experience Required
- 2 to 6 Years
Key Skills
- Breakdown maintenance
- Preventive maintenance
- Vial filling line maintenance
- Lyophilizer handling
- PFS equipment maintenance
- Autoclave maintenance
QMS Department
Available Positions
- Officer
- Senior Officer
Experience Required
- 2 to 6 Years
Key Skills
- SOP preparation
- CAPA handling
- Deviation management
- Change control documentation
- Risk assessment
- Trackwise system knowledge
- Sterile manufacturing compliance
Engineering Department – OSD & Utilities
Utility Operations
Available Positions
- Senior Officer
- Executive
Experience Required
- 4 to 8 Years
Key Skills
- Chiller systems
- Compressors
- Water systems
- Electrical systems
- DG set handling
- GMP compliance
Sterile Manufacturing Department
Available Positions
- Senior Executive
- Assistant Manager
Experience Required
- 8 to 12 Years
Key Skills
- Sterile vial manufacturing
- PFS manufacturing
- Ophthalmic manufacturing
- Lyophilizer operations
- Sterile injectables production
Quality Control Department
QC Microbiology – Sterility / EM
Available Positions
- Senior Officer
- Executive
Experience Required
- 4 to 8 Years
Key Skills
- Sterility testing
- Environmental monitoring
- MLT
- BET testing
Candidates must have experience in USFDA-approved sterile manufacturing facilities.
Finished Goods / Stability Department
Available Positions
- Senior Executive
Experience Required
- 8 to 10 Years
Key Skills
- HPLC analysis
- Dissolution testing
- FTIR analysis
- Finished goods testing
- Stability analysis
Raw Material Quality Control
Available Positions
- Executive
- Assistant Manager
- Officer
- Senior Officer
Experience Required
- 2 to 12 Years
Key Skills
- Raw material sampling
- HPLC testing
- GC testing
- FTIR analysis
- FTNIR operations
- Polarimeter handling
- Auto titrator usage
Candidates with expertise in USFDA-approved OSD formulation facilities may receive additional preference.
Eligibility / Qualifications
Candidates applying for these pharmaceutical vacancies should possess relevant qualifications and regulated manufacturing experience.
Required Educational Qualification
Eligible qualifications include:
- B.Pharm
- M.Pharm
- M.Sc
- ITI
- Diploma
- B.E
- B.Tech
Relevant Courses
B.Pharm, M.Pharm, Pharmaceutical Sciences, Industrial Pharmacy, Pharmaceutical Technology, Microbiology, Clinical Research, Regulatory Affairs, Quality Assurance, Quality Control, Biotechnology, Mechanical Engineering, Electrical Engineering, Industrial Engineering, Sterile Manufacturing, OSD Formulation
Experience Requirement
- Minimum 2 to 12 years of pharmaceutical industry experience
- Experience in USFDA and MHRA-approved facilities is mandatory for several positions
- Candidates should have stability in their current organization for at least 2 years
Preferred Skills
- Strong GMP knowledge
- Regulatory compliance understanding
- Sterile manufacturing exposure
- Pharmaceutical documentation skills
- Team coordination abilities
- Problem-solving and technical troubleshooting skills
- Audit readiness and compliance handling
Candidates looking for pharma company jobs in Daman, sterile injectable manufacturing jobs, QC microbiology careers, regulatory pharmaceutical manufacturing jobs, engineering jobs in pharma plants, and USFDA-approved facility vacancies can attend this walk-in interview.
Location & Salary
Job Location
Daman
Salary
Salary will be offered based on qualifications, technical expertise, regulatory exposure, and pharmaceutical industry experience.
Application Process
Interested candidates can directly attend the walk-in interview with required documents.
Walk-In Interview Details
- Date: 10th May 2026 (Sunday)
- Time: 09:00 AM to 03:00 PM
Walk-In Interview Venue
Baroda Productivity Council, Productivity House, Alkapuri, Vadodara
Documents Required
Candidates attending the interview should carry:
- Updated CV/Resume
- Aadhaar Card
- Passport Size Photographs
- Salary Slips
- Current CTC Breakup
- Educational Certificates
Applicant Prerequisites
- Candidates must have exposure to USFDA and MHRA audits
- Minimum 2 years of stability in the current organization is required
Applicants are advised to arrive early at the venue to complete interview formalities smoothly.
FAQs
1. What is the interview date for USV walk-in recruitment?
The walk-in interview is scheduled for 10th May 2026.
2. What qualifications are required for USV pharma jobs?
Candidates with B.Pharm, M.Pharm, M.Sc, ITI, Diploma, B.E, and B.Tech qualifications can apply.
3. What is the required experience for these vacancies?
Candidates should have 2 to 12 years of pharmaceutical industry experience depending on the position.
4. Which departments are hiring at USV?
USV is hiring for Operations, Engineering, QMS, Sterile Manufacturing, Quality Control, QC Microbiology, Utilities, and Raw Material Testing departments.
5. What is the job location?
The selected candidates will work at the company’s Daman facility.
6. Is experience in USFDA-approved facilities mandatory?
Yes. Several positions require experience in USFDA and MHRA-approved pharmaceutical manufacturing facilities.
Summary Table
| Company | USV Private Limited |
| Department Vacancies | Operations, Engineering, QMS, Sterile Manufacturing, QC, QC Microbiology, Utilities, Raw Material Testing |
| Qualification | B.Pharm, M.Pharm, M.Sc, ITI, Diploma, B.E, B.Tech |
| Experience | 2 to 12 Years |
| Location | Daman |