Stallion Walk-In QA QC Production

- Company Overview
- Job Role & Responsibilities
- Production Department
- Key Skills
- Key Responsibilities
- Warehouse Department
- Key Skills
- Key Responsibilities
- Packing Department
- Key Skills
- Key Responsibilities
- Quality Control Department
- Key Skills
- Key Responsibilities
- Engineering Department
- Key Skills
- Key Responsibilities
- Quality Assurance Department
- Key Skills
- Key Responsibilities
- Formulation & Development (F&D)
- Key Responsibilities
- ADL Department
- Key Skills
- Supply Chain / PPMC Department
- Key Responsibilities
- IT Department
- Key Responsibilities
- Eligibility / Qualifications
- Required Educational Qualification
- Relevant Courses
- Experience Requirement
- Preferred Skills
- Location & Salary
- Job Location
- Salary
- Application Process
- Walk-In Interview Details
- Interview Venue
- Apply Through Email
- Contact Numbers
- Important Instructions
- FAQs
- 1. What is the interview date for Stallion Laboratories recruitment?
- 2. What qualifications are required for these pharma jobs?
- 3. Is USFDA facility experience mandatory?
- 4. Which departments are hiring at Stallion Laboratories?
- 5. What is the required experience for these vacancies?
- 6. How can candidates apply for these openings?
- Summary Table
Stallion Labs Walk-In | QA QC Production Jobs
Stallion Laboratories hiring for QA, QC, Production, Warehouse & Packing. B.Pharm, M.Pharm, ITI eligible in Ahmedabad.
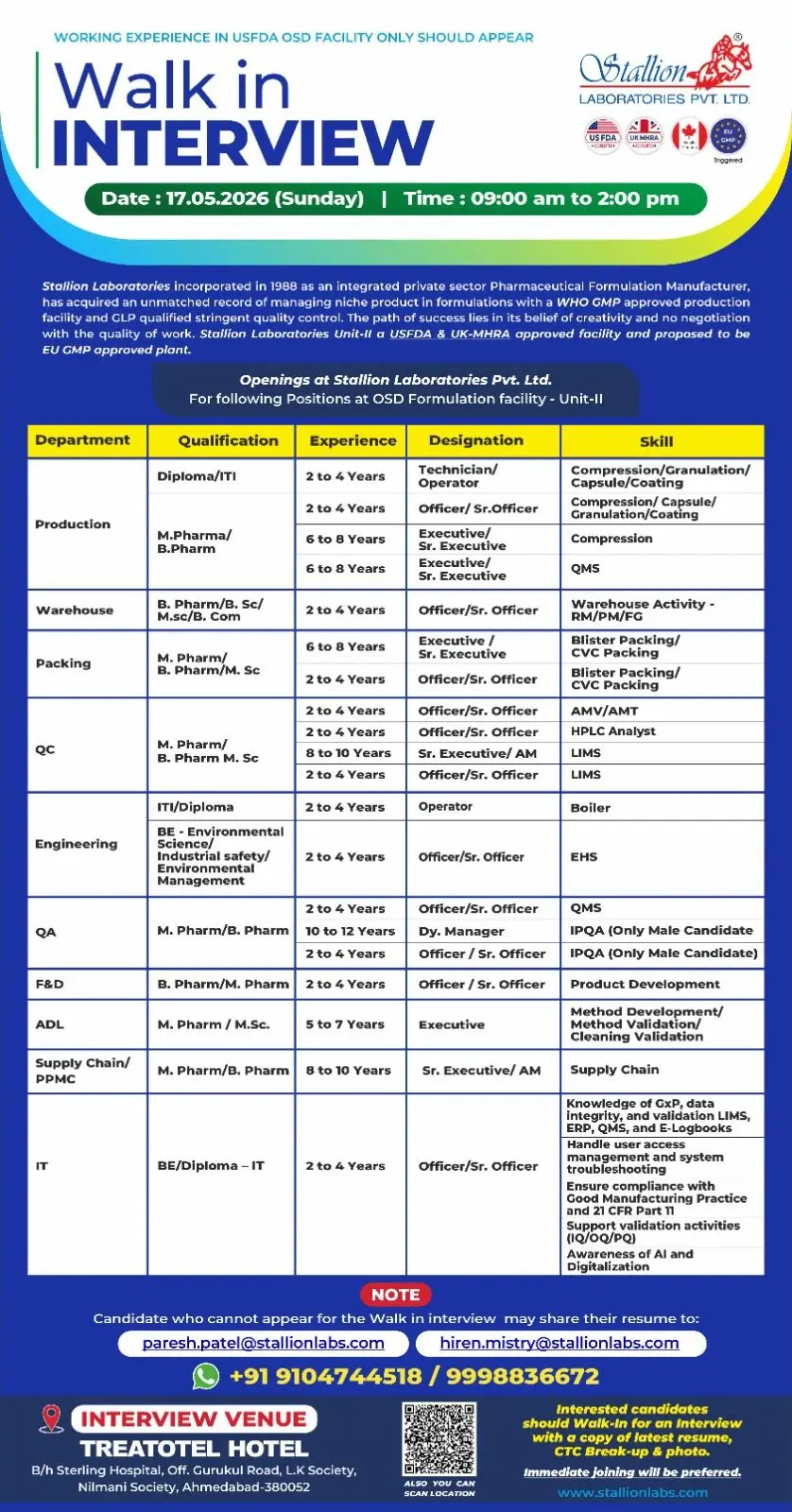
Stallion Laboratories Pvt. Ltd. has announced a walk-in interview drive for experienced pharmaceutical professionals at its USFDA and UK-MHRA approved OSD formulation facility. Candidates searching for pharma manufacturing jobs, QA vacancies, QC analyst openings, production officer careers, warehouse pharma jobs, formulation development opportunities, and regulatory pharmaceutical company careers can attend this latest recruitment drive.
The company is hiring skilled professionals across Production, Packing, Quality Control, Quality Assurance, Warehouse, Engineering, F&D, ADL, Supply Chain, and IT departments. Candidates with experience in USFDA-approved OSD facilities and expertise in regulated pharmaceutical manufacturing operations are encouraged to apply.
This recruitment drive is suitable for professionals with knowledge of cGMP, GxP, data integrity, validation systems, LIMS, ERP, and regulated pharmaceutical manufacturing environments.
Company Overview
Stallion Laboratories Pvt. Ltd. was established in 1988 as an integrated pharmaceutical formulation manufacturing company focused on delivering quality healthcare products for domestic and international markets. The company operates WHO-GMP approved manufacturing facilities with strong expertise in pharmaceutical formulation manufacturing, quality systems, and regulated production operations.
Stallion Laboratories Unit-II is approved by USFDA and UK-MHRA and is also proposed for EU-GMP approval. The organization is recognized for maintaining stringent quality standards, GLP-compliant analytical systems, and advanced pharmaceutical manufacturing practices.
The company continues to strengthen its pharmaceutical operations by recruiting experienced professionals in OSD manufacturing, quality assurance, quality control, formulation development, engineering, and warehouse management.
Job Role & Responsibilities
Production Department
Available Positions
- Technician
- Operator
- Officer
- Senior Officer
- Executive
- Senior Executive
Qualification Required
- Diploma
- ITI
- B.Pharm
- M.Pharm
Experience Required
- 2 to 8 Years
Key Skills
- Compression
- Granulation
- Capsule filling
- Coating operations
- OSD manufacturing processes
Key Responsibilities
- Handle OSD manufacturing operations
- Operate compression, granulation, capsule, and coating equipment
- Maintain compliance with SOPs and cGMP standards
- Ensure production documentation and batch record maintenance
- Support regulated pharmaceutical manufacturing activities
Warehouse Department
Available Positions
- Officer
- Senior Officer
- Executive
- Senior Executive
Qualification Required
- B.Pharm
- B.Sc
- M.Sc
- B.Com
Experience Required
- 2 to 8 Years
Key Skills
- Warehouse RM/PM/FG activities
- Inventory handling
- Material management
Key Responsibilities
- Handle raw material, packing material, and finished goods inventory
- Maintain warehouse documentation and stock records
- Ensure GMP-compliant storage operations
- Coordinate dispensing and material movement activities
Packing Department
Available Positions
- Officer
- Senior Officer
Qualification Required
- M.Pharm
- B.Pharm
- M.Sc
Experience Required
- 2 to 4 Years
Key Skills
- Blister packing
- CVC packing
Key Responsibilities
- Execute pharmaceutical packing operations
- Operate blister and CVC packing machines
- Maintain packaging documentation and cGMP compliance
- Ensure proper track-and-trace and packing process control
Quality Control Department
Available Positions
- Officer
- Senior Officer
- Senior Executive
- Assistant Manager
Qualification Required
- M.Pharm
- B.Pharm
- M.Sc
Experience Required
- 2 to 10 Years
Key Skills
- HPLC analysis
- AMV and AMT
- LIMS operations
Key Responsibilities
- Perform analytical testing using HPLC instruments
- Handle AMV and AMT activities
- Maintain LIMS documentation and compliance
- Support analytical investigations and laboratory quality systems
- Ensure regulatory compliance in analytical operations
Engineering Department
Available Positions
- Operator
- Officer
- Senior Officer
Qualification Required
- ITI
- Diploma
- BE Environmental Science
- Industrial Safety
- Environmental Management
Experience Required
- 2 to 4 Years
Key Skills
- Boiler operations
- EHS management
Key Responsibilities
- Handle engineering utilities and equipment operations
- Support boiler operations and preventive maintenance
- Maintain EHS compliance and workplace safety systems
- Ensure environmental and industrial safety standards
Quality Assurance Department
Available Positions
- Deputy Manager
- Officer
- Senior Officer
Qualification Required
- M.Pharm
- B.Pharm
Experience Required
- 2 to 12 Years
Key Skills
- QMS
- IPQA
- Documentation systems
Key Responsibilities
- Handle QMS documentation and compliance activities
- Perform IPQA monitoring and manufacturing checks
- Maintain GMP and regulatory documentation systems
- Support audits, deviations, and CAPA activities
Formulation & Development (F&D)
Available Positions
- Officer
- Senior Officer
Qualification Required
- B.Pharm
- M.Pharm
Experience Required
- 2 to 4 Years
Key Responsibilities
- Support product development activities
- Handle formulation development documentation
- Coordinate product evaluation and manufacturing support
ADL Department
Available Positions
- Executive
Qualification Required
- M.Pharm
- M.Sc
Experience Required
- 5 to 7 Years
Key Skills
- Method development
- Method validation
- Cleaning validation
Supply Chain / PPMC Department
Available Positions
- Senior Executive
- Assistant Manager
Qualification Required
- M.Pharm
- B.Pharm
Experience Required
- 8 to 10 Years
Key Responsibilities
- Manage supply chain activities
- Coordinate production planning and material control
- Support inventory and logistics operations
IT Department
Available Positions
- Officer
- Senior Officer
Qualification Required
- BE IT
- Diploma IT
Experience Required
- 2 to 4 Years
Key Responsibilities
- Handle LIMS, ERP, QMS, and E-logbook systems
- Manage user access and system troubleshooting
- Support validation activities including IQ, OQ, and PQ
- Maintain compliance with 21 CFR Part 11 guidelines
- Support digitalization and AI awareness initiatives
Eligibility / Qualifications
Candidates applying for these pharmaceutical vacancies should possess relevant educational qualifications and experience in regulated pharmaceutical manufacturing facilities.
Required Educational Qualification
Eligible qualifications include:
- ITI
- Diploma
- B.Pharm
- M.Pharm
- B.Sc
- M.Sc
- B.Com
- BE IT
- BE Environmental Science
Relevant Courses
B.Pharm, M.Pharm, Pharmaceutical Sciences, Industrial Pharmacy, Pharmaceutical Technology, Regulatory Affairs, Clinical Research, Analytical Chemistry, Biotechnology, Environmental Science, Industrial Safety, Information Technology, Inventory Management, Quality Assurance, Quality Control, Formulation Development, Supply Chain Management
Experience Requirement
- Minimum 2 to 12 years of pharmaceutical industry experience
- Experience in USFDA-approved OSD facilities is mandatory
- Candidates with exposure to UK-MHRA and EU-GMP facilities may receive preference
Preferred Skills
- cGMP and GxP compliance knowledge
- Data integrity and validation systems understanding
- LIMS, ERP, QMS, and E-logbook handling
- Pharmaceutical manufacturing documentation expertise
- Analytical testing and HPLC handling skills
- Warehouse and inventory management knowledge
- Problem-solving and communication abilities
Candidates searching for pharma walk-in interviews in Ahmedabad, QA manager jobs, QC analyst openings, OSD manufacturing careers, formulation development jobs, and warehouse pharmaceutical vacancies can attend this recruitment drive.
Location & Salary
Job Location
Ahmedabad
Salary
Salary will be offered according to candidate qualifications, pharmaceutical industry experience, technical expertise, and company standards.
Application Process
Interested and eligible candidates can directly attend the walk-in interview or send their updated resume through email.
Walk-In Interview Details
- Date: 17th May 2026
- Time: 09:00 AM to 02:00 PM
Interview Venue
Treatotel Hotel Behind Sterling Hospital, Off Gurukul Road, L.K Society, Nilmani Society, Ahmedabad – 380052
Apply Through Email
Contact Numbers
- +91 9104744518
- +91 9998836672
Important Instructions
Candidates attending the interview should carry:
- Updated Resume/CV
- Passport Size Photograph
- Latest CTC Breakup
- Educational Certificates
- Experience Certificates
- Government ID Proof
Immediate joiners may receive preference during the recruitment process.
FAQs
1. What is the interview date for Stallion Laboratories recruitment?
The walk-in interview is scheduled for 17th May 2026.
2. What qualifications are required for these pharma jobs?
Candidates with ITI, Diploma, B.Pharm, M.Pharm, B.Sc, M.Sc, B.Com, and BE qualifications can apply.
3. Is USFDA facility experience mandatory?
Yes. Candidates with working experience in USFDA-approved OSD facilities should apply.
4. Which departments are hiring at Stallion Laboratories?
The company is hiring for Production, Warehouse, Packing, QC, QA, Engineering, F&D, ADL, Supply Chain, and IT departments.
5. What is the required experience for these vacancies?
Candidates should have 2 to 12 years of pharmaceutical industry experience.
6. How can candidates apply for these openings?
Candidates can attend the walk-in interview directly or send their updated resume to the provided email IDs.
Summary Table
| Company | Stallion Laboratories Pvt. Ltd. |
| Department Vacancies | Production, Warehouse, Packing, QC, QA, Engineering, F&D, ADL, Supply Chain, IT |
| Qualification | ITI, Diploma, B.Pharm, M.Pharm, B.Sc, M.Sc, B.Com, BE |
| Experience | 2 to 12 Years |
| Location | Ahmedabad, Gujarat |
You must sign in to apply for this position.


