Ipca Walk-In QA QC Production

- Company Overview
- Job Role & Responsibilities
- Microbiology Department
- Learning Opportunities
- Quality Control Department – Analytical, Molecular Biology & Bioassay
- Learning Opportunities
- Quality Control Department – RM / PM
- Learning Opportunities
- QMS, Reviewer & Compliance Department
- Learning Opportunities
- Quality Assurance Department
- Learning Opportunities
- Production Department – DS Manufacturing / DP Manufacturing
- Learning Opportunities
- Eligibility / Qualifications
- Required Educational Qualification
- Relevant Courses
- Experience Requirement
- Preferred Skills
- Location & Salary
- Job Location
- Salary
- Application Process
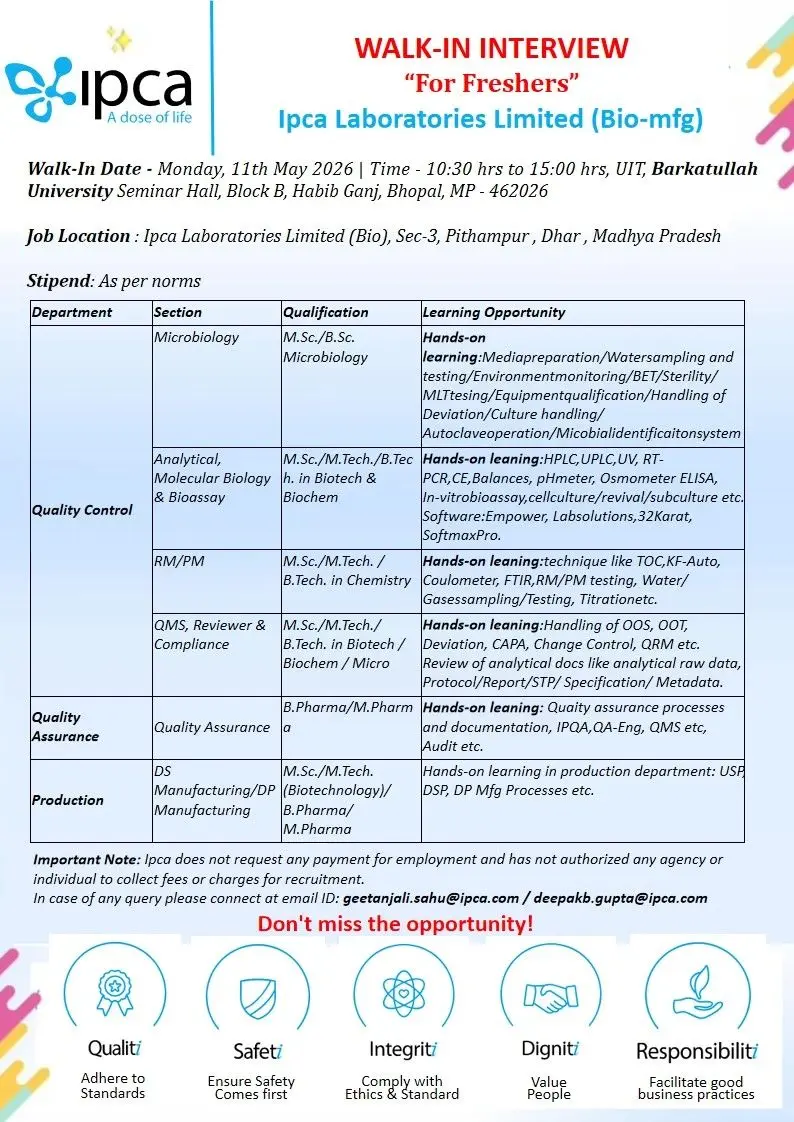
- Walk-In Interview Details
- Interview Venue
- Job Location
- Contact Email IDs
- Important Note
- Documents Required
- FAQs
- 1. Who can apply for Ipca Laboratories fresher recruitment?
- 2. What departments are hiring at Ipca Laboratories?
- 3. What is the interview date for the Ipca walk-in drive?
- 4. What is the job location?
- 5. Are freshers eligible for this recruitment drive?
- 6. What learning opportunities are provided during training?
- Summary Table
Ipca Labs Freshers Walk-In | QA QC Production Jobs
Ipca Laboratories hiring freshers for QA, QC, Production & Microbiology. B.Sc, M.Sc, B.Pharm eligible in Pithampur.
Ipca Laboratories Limited has announced a walk-in interview drive for freshers at its Bio-manufacturing facility located in Pithampur, Madhya Pradesh. Candidates searching for pharma fresher jobs, biotechnology careers, microbiology trainee openings, quality control jobs for freshers, production trainee opportunities, and pharmaceutical industry walk-in interviews can attend this latest recruitment drive.
The company is offering learning and training opportunities across Microbiology, Quality Control, Quality Assurance, QMS, Compliance, and Production departments for fresh graduates and postgraduates from biotechnology, microbiology, pharmacy, chemistry, and life sciences backgrounds.
Candidates with B.Sc, M.Sc, B.Tech, M.Tech, B.Pharm, and M.Pharm qualifications who are interested in pharmaceutical manufacturing, analytical laboratories, biotechnology production, and healthcare quality systems can explore this fresher hiring opportunity.
Company Overview
Ipca Laboratories Limited is one of India’s leading pharmaceutical companies engaged in manufacturing and supplying high-quality pharmaceutical and healthcare products across domestic and international markets. The company operates advanced pharmaceutical manufacturing and biotechnology facilities with strong expertise in quality systems, research-driven healthcare solutions, and regulated manufacturing operations.
The organization is known for its focus on quality standards, ethics, innovation, safety, and employee development. Ipca continues to invest in pharmaceutical manufacturing, biotechnology operations, analytical laboratories, and healthcare product development.
The company offers excellent learning opportunities for freshers who want to build careers in pharmaceutical manufacturing, microbiology, biotechnology research, analytical quality control, and healthcare compliance operations.
Job Role & Responsibilities
Microbiology Department
Qualification Required
- M.Sc Microbiology
- B.Sc Microbiology
Learning Opportunities
Selected candidates will receive practical exposure in:
- Media preparation
- Water sampling and testing
- Environmental monitoring
- BET testing
- Sterility testing
- MLT testing
- Equipment qualification
- Handling deviations
- Culture handling
- Autoclave operations
- Microbial identification systems
Quality Control Department – Analytical, Molecular Biology & Bioassay
Qualification Required
- M.Sc Biotechnology
- M.Tech Biotechnology
- B.Tech Biotechnology
- M.Sc Biochemistry
Learning Opportunities
Candidates will gain hands-on exposure in:
- HPLC operations
- UPLC analysis
- UV spectroscopy
- RT-PCR techniques
- Capillary electrophoresis
- ELISA testing
- Osmometer handling
- Cell culture and subculture
- Analytical software including Empower, LabSolutions, 32 Karat, and SoftmaxPro
Quality Control Department – RM / PM
Qualification Required
- M.Sc Chemistry
- M.Tech Chemistry
- B.Tech Chemistry
Learning Opportunities
Selected candidates will receive practical exposure in:
- TOC analysis
- KF Auto and Coulometer operations
- FTIR analysis
- RM and PM testing
- Water and gas sampling
- Titration techniques
- Pharmaceutical analytical testing procedures
QMS, Reviewer & Compliance Department
Qualification Required
- M.Sc Biotechnology
- M.Tech Biotechnology
- B.Tech Biotechnology
- M.Sc Biochemistry
- M.Sc Microbiology
Learning Opportunities
Candidates will gain practical exposure in:
- Handling OOS and OOT investigations
- Deviation and CAPA management
- Change control systems
- Quality risk management (QRM)
- Review of analytical documents
- Protocol, report, and specification review
- Metadata and raw data handling
Quality Assurance Department
Qualification Required
- B.Pharm
- M.Pharm
Learning Opportunities
Candidates will receive practical training in:
- IPQA activities
- QA engineering systems
- Quality management systems
- Audit handling and documentation
- Pharmaceutical quality assurance procedures
- GMP compliance activities
Production Department – DS Manufacturing / DP Manufacturing
Qualification Required
- M.Sc Biotechnology
- M.Tech Biotechnology
- B.Pharm
- M.Pharm Biotechnology
Learning Opportunities
Selected candidates will gain practical manufacturing exposure in:
- USP operations
- DSP operations
- Drug substance manufacturing
- Drug product manufacturing
- Biotechnology production processes
- Pharmaceutical manufacturing systems
Eligibility / Qualifications
Candidates applying for these fresher pharmaceutical vacancies should possess relevant educational qualifications from life sciences, biotechnology, pharmacy, and healthcare backgrounds.
Required Educational Qualification
Eligible qualifications include:
- B.Sc
- M.Sc
- B.Tech
- M.Tech
- B.Pharm
- M.Pharm
Relevant Courses
B.Sc Microbiology, M.Sc Microbiology, Biotechnology, Biochemistry, Pharmaceutical Sciences, B.Pharm, M.Pharm Biotechnology, Molecular Biology, Clinical Research, Pharmaceutical Analysis, Quality Assurance, Quality Control, Life Sciences, Analytical Chemistry, Biotechnology Engineering, Healthcare Sciences
Experience Requirement
- Freshers are eligible to apply
- Candidates interested in pharmaceutical manufacturing and analytical laboratories may receive preference
- Strong academic background in biotechnology, pharmacy, microbiology, or chemistry preferred
Preferred Skills
- Basic understanding of pharmaceutical quality systems
- Knowledge of analytical instruments and laboratory techniques
- Communication and teamwork abilities
- Willingness to learn pharmaceutical manufacturing operations
- Documentation and compliance awareness
- Problem-solving and analytical thinking capabilities
Candidates searching for pharma jobs for freshers, biotechnology walk-in interviews, microbiology trainee jobs, QA fresher openings, analytical laboratory careers, and biotechnology manufacturing opportunities can attend this recruitment drive.
Location & Salary
Job Location
Pithampur
Salary
Stipend will be offered according to company norms and training structure.
Application Process
Interested and eligible candidates can directly attend the walk-in interview drive at the mentioned venue.
Walk-In Interview Details
- Date: Monday, 11th May 2026
- Time: 10:30 AM to 03:00 PM
Interview Venue
UIT, Barkatullah University Seminar Hall, Block B, Habib Ganj, Bhopal, Madhya Pradesh – 462026
Job Location
Ipca Laboratories Limited (Bio) Sector-3, Pithampur, Dhar, Madhya Pradesh
Contact Email IDs
Important Note
Ipca Laboratories Limited does not request any payment for employment opportunities and has not authorized any agency or individual to collect recruitment fees from candidates.
Documents Required
Candidates attending the interview should carry:
- Updated Resume/CV
- Educational Certificates
- Government ID Proof
- Passport Size Photographs
- Academic Project Details
Applicants are encouraged to attend the interview on time to complete the recruitment process smoothly.
FAQs
1. Who can apply for Ipca Laboratories fresher recruitment?
Freshers with B.Sc, M.Sc, B.Tech, M.Tech, B.Pharm, and M.Pharm qualifications can apply.
2. What departments are hiring at Ipca Laboratories?
The company is hiring for Microbiology, Quality Control, Quality Assurance, QMS, Compliance, and Production departments.
3. What is the interview date for the Ipca walk-in drive?
The walk-in interview is scheduled for 11th May 2026.
4. What is the job location?
The selected candidates will work at Ipca Laboratories Bio-manufacturing facility in Pithampur, Madhya Pradesh.
5. Are freshers eligible for this recruitment drive?
Yes. This walk-in drive is specially organized for freshers.
6. What learning opportunities are provided during training?
Candidates will receive hands-on exposure in microbiology testing, HPLC, RT-PCR, QA systems, biotechnology manufacturing, analytical laboratories, and pharmaceutical quality systems.
Summary Table
| Company | Ipca Laboratories Limited |
| Department Vacancies | Microbiology, Quality Control, Quality Assurance, QMS, Compliance, Production |
| Qualification | B.Sc, M.Sc, B.Tech, M.Tech, B.Pharm, M.Pharm |
| Experience | Freshers |
| Location | Pithampur, Madhya Pradesh |
You must sign in to apply for this position.


