Lactose Hiring QA QC Production

- Company Overview
- Job Role & Responsibilities
- Quality Control Department
- Key Responsibilities
- Quality Assurance Department
- Key Responsibilities
- Store Department
- Key Responsibilities
- Production Department
- Key Responsibilities
- Production API Department
- Key Responsibilities
- Production OSD Department
- Key Responsibilities
- Project & Engineering Department
- Key Responsibilities
- SRM Department
- Key Responsibilities
- Process Development Department
- Key Responsibilities
- Eligibility / Qualifications
- Required Educational Qualification
- Relevant Courses
- Experience Requirement
- Preferred Skills
- Location & Salary
- Job Location
- Salary
- Application Process
- Apply Through Email
- Manufacturing Facility Location
- Documents Required
- FAQs
- 1. Which departments are hiring at Lactose India Limited?
- 2. What qualifications are required for these pharma jobs?
- 3. What is the required experience for these vacancies?
- 4. What is the job location?
- 5. Are engineering candidates eligible for these openings?
- 6. How can candidates apply for these vacancies?
- Summary Table
Lactose India Hiring | QA QC Production Jobs
Lactose India hiring for QA, QC, Production, Engineering & Store roles. B.Pharm, M.Pharm, B.Sc eligible in Vadodara.
Lactose India Limited has announced multiple pharmaceutical and engineering job openings at its manufacturing facility located in Poicha (Rania), Vadodara. Candidates searching for pharma manufacturing jobs, API production careers, quality assurance vacancies, quality control jobs, engineering positions, process development openings, and pharmaceutical company opportunities in Gujarat can apply for these latest recruitment opportunities.
The company is hiring experienced professionals across Quality Control, Quality Assurance, Production API, Production OSD, Store, Engineering, SRM, Process Development, and Project departments. Candidates with B.Sc, M.Sc, B.Pharm, M.Pharm, B.E, M.E, Diploma, and environmental science qualifications who are looking for regulated pharmaceutical manufacturing careers can explore these openings.
This recruitment drive is suitable for professionals seeking growth in pharmaceutical manufacturing, process development, engineering maintenance, quality systems, inventory management, and healthcare production operations.
Company Overview
Lactose India Limited is a pharmaceutical manufacturing company engaged in producing healthcare and pharmaceutical products with a focus on quality manufacturing standards and industrial excellence. The company operates manufacturing facilities supporting pharmaceutical production, API manufacturing, OSD operations, engineering systems, and regulated quality processes.
The organization continues to strengthen its workforce by recruiting skilled pharmaceutical professionals, engineering experts, production specialists, environmental science professionals, and quality assurance teams to support its manufacturing expansion.
Lactose India Limited provides career growth opportunities for candidates involved in pharmaceutical manufacturing operations, regulatory compliance, process optimization, engineering management, and quality systems.
Job Role & Responsibilities
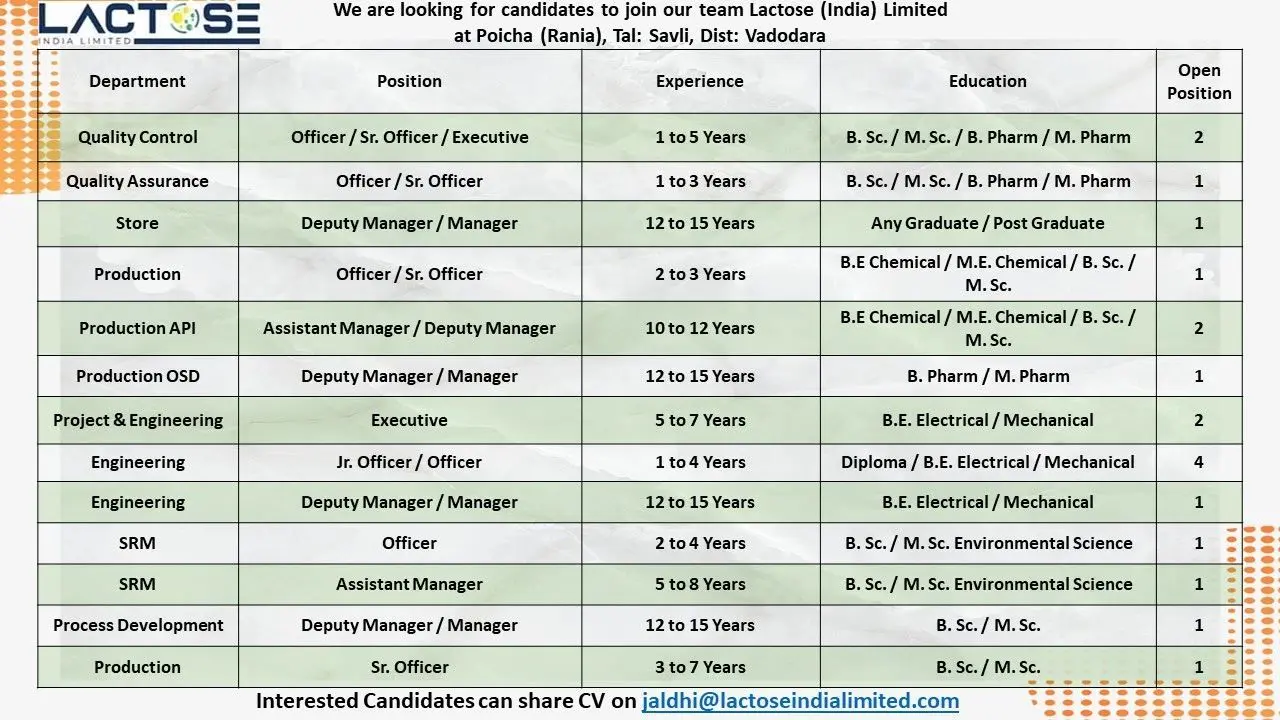
Quality Control Department
Available Positions
- Officer
- Senior Officer
- Executive
Open Positions
- 2
Qualification Required
- B.Sc
- M.Sc
- B.Pharm
- M.Pharm
Experience Required
- 1 to 5 Years
Key Responsibilities
- Perform analytical testing of pharmaceutical products and raw materials
- Maintain laboratory documentation and GMP compliance
- Handle quality testing procedures and analytical investigations
- Support regulatory and quality compliance activities
- Ensure accurate reporting of laboratory results
Quality Assurance Department
Available Positions
- Officer
- Senior Officer
Open Positions
- 1
Qualification Required
- B.Sc
- M.Sc
- B.Pharm
- M.Pharm
Experience Required
- 1 to 3 Years
Key Responsibilities
- Maintain quality assurance documentation and compliance records
- Support GMP and SOP implementation activities
- Conduct quality checks and process monitoring
- Participate in audits and quality investigations
Store Department
Available Positions
- Deputy Manager
- Manager
Open Positions
- 1
Qualification Required
- Any Graduate
- Post Graduate
Experience Required
- 12 to 15 Years
Key Responsibilities
- Manage pharmaceutical inventory and warehouse operations
- Maintain material handling and stock records
- Coordinate raw material and finished goods movement
- Ensure efficient storage and inventory management practices
Production Department
Available Positions
- Officer
- Senior Officer
Open Positions
- 1
Qualification Required
- B.E Chemical
- M.E Chemical
- B.Sc
- M.Sc
Experience Required
- 2 to 3 Years
Key Responsibilities
- Handle pharmaceutical manufacturing operations
- Monitor production process efficiency and compliance
- Maintain batch manufacturing documentation
- Ensure safe and compliant manufacturing activities
Production API Department
Available Positions
- Assistant Manager
- Deputy Manager
Open Positions
- 2
Qualification Required
- B.E Chemical
- M.E Chemical
- B.Sc
- M.Sc
Experience Required
- 10 to 12 Years
Key Responsibilities
- Manage API production activities and process operations
- Supervise manufacturing teams and production schedules
- Maintain GMP compliance and process safety standards
- Support process optimization and production efficiency
Production OSD Department
Available Positions
- Deputy Manager
- Manager
Open Positions
- 1
Qualification Required
- B.Pharm
- M.Pharm
Experience Required
- 12 to 15 Years
Key Responsibilities
- Handle OSD manufacturing operations and team management
- Ensure compliance with pharmaceutical manufacturing standards
- Monitor production activities and documentation systems
- Coordinate manufacturing process improvement initiatives
Project & Engineering Department
Available Positions
- Executive
- Junior Officer
- Officer
- Deputy Manager
- Manager
Open Positions
- 7
Qualification Required
- B.E Electrical
- B.E Mechanical
- Diploma Electrical
- Diploma Mechanical
Experience Required
- 1 to 15 Years
Key Responsibilities
- Handle engineering maintenance and project activities
- Support electrical and mechanical systems operations
- Ensure preventive maintenance and equipment reliability
- Maintain engineering compliance and safety standards
SRM Department
Available Positions
- Officer
- Assistant Manager
Open Positions
- 2
Qualification Required
- B.Sc Environmental Science
- M.Sc Environmental Science
Experience Required
- 2 to 8 Years
Key Responsibilities
- Handle environmental compliance and sustainability activities
- Monitor environmental safety standards and reporting systems
- Support environmental management initiatives
- Maintain regulatory compliance documentation
Process Development Department
Available Positions
- Deputy Manager
- Manager
Open Positions
- 1
Qualification Required
- B.Sc
- M.Sc
Experience Required
- 12 to 15 Years
Key Responsibilities
- Support pharmaceutical process development activities
- Handle process optimization and manufacturing improvements
- Coordinate development projects and documentation systems
- Maintain process validation and compliance activities
Eligibility / Qualifications
Candidates applying for these pharmaceutical vacancies should possess relevant educational qualifications and regulated manufacturing experience.
Required Educational Qualification
Eligible qualifications include:
- B.Sc
- M.Sc
- B.Pharm
- M.Pharm
- B.E Chemical
- M.E Chemical
- B.E Electrical
- B.E Mechanical
- Diploma
- Environmental Science
- Graduate
- Post Graduate
Relevant Courses
B.Pharm, M.Pharm, Pharmaceutical Sciences, Industrial Pharmacy, Pharmaceutical Technology, Chemical Engineering, Mechanical Engineering, Electrical Engineering, Environmental Science, Biotechnology, Quality Assurance, Quality Control, Regulatory Affairs, Clinical Research, Process Development, Inventory Management, Manufacturing Operations
Experience Requirement
- Minimum 1 to 15 years of pharmaceutical industry experience
- Candidates with manufacturing and regulatory exposure may receive preference
- Experience in API, OSD, engineering, and pharmaceutical production operations preferred
Preferred Skills
- GMP and regulatory compliance understanding
- Pharmaceutical manufacturing documentation knowledge
- Analytical laboratory and quality systems expertise
- Engineering maintenance and troubleshooting skills
- Process optimization and production management abilities
- Inventory and warehouse management exposure
- Team coordination and communication skills
Candidates searching for pharma jobs in Vadodara, API manufacturing careers, QA QC vacancies, pharmaceutical engineering jobs, process development openings, and production manager positions can apply for these opportunities.
Location & Salary
Job Location
Vadodara
Salary
Salary will be offered according to candidate qualifications, pharmaceutical industry experience, technical expertise, and company standards.
Application Process
Interested and eligible candidates can apply by sharing their updated resume through email.
Apply Through Email
Manufacturing Facility Location
Lactose India Limited Poicha (Rania), Taluka Savli, District Vadodara, Gujarat
Documents Required
Candidates applying for these vacancies should keep the following documents ready:
- Updated Resume/CV
- Educational Certificates
- Experience Certificates
- Government ID Proof
- Latest Salary Details
- Passport Size Photographs
Applicants are encouraged to apply early because shortlisting may begin based on relevant pharmaceutical and engineering experience.
FAQs
1. Which departments are hiring at Lactose India Limited?
The company is hiring for Quality Control, Quality Assurance, Production API, Production OSD, Store, Engineering, SRM, and Process Development departments.
2. What qualifications are required for these pharma jobs?
Candidates with B.Sc, M.Sc, B.Pharm, M.Pharm, B.E, M.E, Diploma, and environmental science qualifications can apply.
3. What is the required experience for these vacancies?
Candidates should have 1 to 15 years of pharmaceutical industry experience depending on the position.
4. What is the job location?
The job location is Poicha (Rania), Vadodara, Gujarat.
5. Are engineering candidates eligible for these openings?
Yes. Candidates with Electrical, Mechanical, and Chemical Engineering qualifications can apply for engineering and production positions.
6. How can candidates apply for these vacancies?
Interested candidates can send their updated resume to jaldhi@lactoseindialimited.com.
Summary Table
| Company | Lactose India Limited |
| Department Vacancies | Quality Control, Quality Assurance, Production API, Production OSD, Store, Engineering, SRM, Process Development |
| Qualification | B.Sc, M.Sc, B.Pharm, M.Pharm, B.E, M.E, Diploma |
| Experience | 1 to 15 Years |
| Location | Vadodara, Gujarat |
You must sign in to apply for this position.


