Macleods Walk-In QA QC Production

- Company Overview
- Job Role & Responsibilities
- Quality Control Department – OSD
- Key Responsibilities
- Quality Control Department – Peptide API
- Key Responsibilities
- Quality Control Department – Injectable Block
- Key Responsibilities
- Production Department – Peptide API
- Key Responsibilities
- Engineering Services Department – Injectable Block
- Key Responsibilities
- Quality Assurance Department – OSD
- Key Responsibilities
- Quality Assurance Department – Injectable Block
- Key Responsibilities
- Eligibility / Qualifications
- Required Educational Qualification
- Relevant Courses
- Experience Requirement
- Preferred Skills
- Location & Salary
- Job Location
- Salary
- Application Process
- Registration Link
- Walk-In Interview Details
- Interview Venue
- Contact Numbers
- Documents Required
- FAQs
- 1. Which departments are hiring at Macleods Pharmaceuticals?
- 2. What qualifications are required for these pharma jobs?
- 3. What is the required experience for these vacancies?
- 4. What are the job locations?
- 5. Is registration mandatory before attending the interview?
- 6. What skills are preferred for these pharmaceutical roles?
- Summary Table
Macleods Walk-In | QA QC Production Jobs
Macleods hiring for QA, QC, Engineering & Peptide Production roles. B.Pharm, M.Pharm eligible in Indore & Daman.
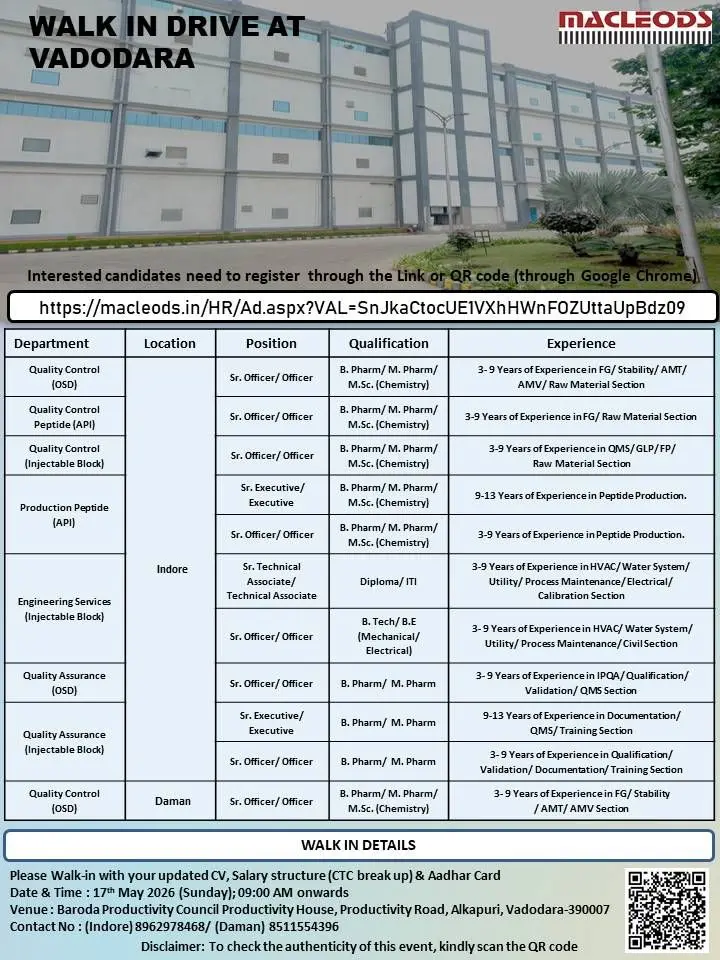
Macleods Pharmaceuticals has announced a walk-in drive in Vadodara for experienced pharmaceutical professionals across Quality Control, Quality Assurance, Production Peptide, and Engineering Services departments. Candidates searching for pharma QA jobs, QC officer vacancies, injectable manufacturing careers, peptide API production jobs, engineering maintenance openings, and pharmaceutical company opportunities can attend this latest recruitment drive.
The company is hiring experienced professionals for its OSD, Injectable, and API manufacturing divisions located in Indore and Daman facilities. Candidates with expertise in regulated pharmaceutical manufacturing, quality systems, engineering services, validation, documentation, and peptide production operations are encouraged to apply.
This recruitment drive is suitable for professionals seeking careers in pharmaceutical manufacturing, quality assurance, quality control, injectable operations, engineering maintenance, peptide production, and healthcare compliance systems.
Company Overview
Macleods Pharmaceuticals is one of India’s leading pharmaceutical companies known for manufacturing high-quality pharmaceutical formulations, APIs, injectables, and healthcare products for domestic and international markets. The company operates advanced pharmaceutical manufacturing facilities that comply with global regulatory standards and quality systems.
The organization focuses on regulated manufacturing operations, GMP compliance, pharmaceutical innovation, and workforce development across multiple healthcare and pharmaceutical divisions.
Macleods continues to expand its pharmaceutical manufacturing operations by recruiting experienced professionals for quality systems, engineering services, peptide production, injectable manufacturing, and pharmaceutical compliance roles.
The company provides strong career growth opportunities for candidates with experience in pharmaceutical production, quality management systems, validation, analytical laboratories, and regulated healthcare manufacturing operations.
Job Role & Responsibilities
Quality Control Department – OSD
Available Positions
- Senior Officer
- Officer
Qualification Required
- B.Pharm
- M.Pharm
- M.Sc Chemistry
Experience Required
- 3 to 9 Years
Key Responsibilities
- Handle finished goods and stability analysis
- Perform AMT and AMV activities
- Support raw material testing operations
- Maintain analytical laboratory documentation and GLP compliance
- Ensure regulatory and GMP compliance in quality control operations
Quality Control Department – Peptide API
Available Positions
- Senior Officer
- Officer
Qualification Required
- B.Pharm
- M.Pharm
- M.Sc Chemistry
Experience Required
- 3 to 9 Years
Key Responsibilities
- Perform raw material and finished goods analysis for peptide API products
- Maintain analytical testing documentation and compliance records
- Support regulated pharmaceutical laboratory activities
- Ensure adherence to quality standards and SOP procedures
Quality Control Department – Injectable Block
Available Positions
- Senior Officer
- Officer
Qualification Required
- B.Pharm
- M.Pharm
- M.Sc Chemistry
Experience Required
- 3 to 9 Years
Key Responsibilities
- Handle QMS, GLP, finished product, and raw material testing activities
- Maintain injectable quality systems and analytical documentation
- Support regulatory audit readiness and laboratory compliance
- Ensure GMP compliance during testing operations
Production Department – Peptide API
Available Positions
- Senior Executive
- Executive
Qualification Required
- B.Pharm
- M.Pharm
- M.Sc Chemistry
Experience Required
- 9 to 13 Years
Key Responsibilities
- Handle peptide API manufacturing operations
- Supervise pharmaceutical production activities and process execution
- Maintain GMP documentation and production compliance systems
- Support batch manufacturing and process optimization activities
Engineering Services Department – Injectable Block
Available Positions
- Senior Technical Associate
- Technical Associate
- Senior Officer
- Officer
Qualification Required
- Diploma
- ITI
- B.Tech Mechanical
- B.Tech Electrical
- B.E Mechanical
- B.E Electrical
Experience Required
- 3 to 9 Years
Key Responsibilities
- Handle HVAC and water system maintenance activities
- Support utility, process maintenance, electrical, and calibration operations
- Maintain engineering documentation and preventive maintenance systems
- Ensure compliance with pharmaceutical engineering standards
- Support civil and facility maintenance operations
Quality Assurance Department – OSD
Available Positions
- Senior Officer
- Officer
- Senior Executive
- Executive
Qualification Required
- B.Pharm
- M.Pharm
Experience Required
- 3 to 13 Years
Key Responsibilities
- Handle IPQA, qualification, and validation activities
- Manage QMS documentation and compliance systems
- Support training and audit activities
- Maintain pharmaceutical documentation and SOP compliance
- Coordinate GMP and regulatory quality assurance operations
Quality Assurance Department – Injectable Block
Available Positions
- Senior Officer
- Officer
Qualification Required
- B.Pharm
- M.Pharm
Experience Required
- 3 to 9 Years
Key Responsibilities
- Support qualification, validation, documentation, and training activities
- Maintain injectable manufacturing quality systems
- Ensure compliance with GMP and regulatory guidelines
- Coordinate quality investigations and documentation reviews
Eligibility / Qualifications
Candidates applying for these pharmaceutical vacancies should possess relevant educational qualifications and regulated pharmaceutical manufacturing experience.
Required Educational Qualification
Eligible qualifications include:
- B.Pharm
- M.Pharm
- M.Sc Chemistry
- Diploma
- ITI
- B.Tech Mechanical
- B.Tech Electrical
- B.E Mechanical
- B.E Electrical
Relevant Courses
B.Pharm, M.Pharm, Pharmaceutical Sciences, Industrial Pharmacy, Pharmaceutical Technology, Analytical Chemistry, Biotechnology, Chemical Analysis, Mechanical Engineering, Electrical Engineering, Process Engineering, Quality Assurance, Quality Control, Validation, Regulatory Compliance, Pharmaceutical Manufacturing
Experience Requirement
- Minimum 3 to 13 years of pharmaceutical industry experience
- Candidates with regulated manufacturing exposure may receive preference
- Experience in OSD, Injectable, API, and peptide manufacturing operations preferred
Preferred Skills
- GMP and GLP compliance understanding
- QMS, AMT, and AMV knowledge
- Validation and qualification expertise
- HVAC and utility maintenance exposure
- Analytical laboratory handling skills
- Pharmaceutical manufacturing documentation experience
- Team coordination and communication abilities
Candidates searching for pharma walk-in interviews in Vadodara, QC officer jobs, QA executive openings, injectable manufacturing careers, engineering pharma jobs, and peptide API production vacancies can attend this recruitment drive.
Location & Salary
Job Location
Indore / Daman
Salary
Salary will be offered according to candidate qualifications, pharmaceutical industry experience, technical expertise, and company standards.
Application Process
Interested and eligible candidates should register through the official registration link before attending the walk-in interview.
Registration Link
https://macleods.in/HR/Ad.aspx?VAL=SnJkaCtocUE1VXhHWnFOZUttaUpBdz09
Walk-In Interview Details
- Date: 17th May 2026
- Day: Sunday
- Time: 09:00 AM onwards
Interview Venue
Baroda Productivity Council Productivity House, Productivity Road, Alkapuri, Vadodara – 390007
Contact Numbers
- Indore: 8962978468
- Daman: 8511554396
Documents Required
Candidates attending the interview should carry:
- Updated Resume/CV
- Latest Salary Structure and CTC Breakup
- Aadhaar Card
- Educational Certificates
- Experience Certificates
- Passport Size Photographs
Applicants are encouraged to register and attend the interview on time to complete the recruitment process smoothly.
FAQs
1. Which departments are hiring at Macleods Pharmaceuticals?
The company is hiring for Quality Control, Quality Assurance, Production Peptide API, and Engineering Services departments.
2. What qualifications are required for these pharma jobs?
Candidates with B.Pharm, M.Pharm, M.Sc Chemistry, Diploma, ITI, B.Tech, and B.E qualifications can apply.
3. What is the required experience for these vacancies?
Candidates should have 3 to 13 years of pharmaceutical industry experience.
4. What are the job locations?
The available job locations are Indore and Daman.
5. Is registration mandatory before attending the interview?
Yes. Candidates should register through the official Macleods registration link before attending the walk-in drive.
6. What skills are preferred for these pharmaceutical roles?
Candidates with expertise in GMP, GLP, validation, HVAC systems, injectable manufacturing, peptide production, and QMS may receive preference.
Summary Table
| Company | Macleods Pharmaceuticals |
| Department Vacancies | Quality Control, Quality Assurance, Production Peptide API, Engineering Services |
| Qualification | B.Pharm, M.Pharm, M.Sc Chemistry, Diploma, ITI, B.Tech, B.E |
| Experience | 3 to 13 Years |
| Location | Indore / Daman |
To apply for this job please visit macleods.in.


