Adley walk-in QC & Production

- Company Overview
- Walk-In Interview Details
- Job Role & Responsibilities
- 1. FG HPLC Analyst – Quality Control (Officer)
- 2. AMV – Analytical Method Validation (Sr. Officer)
- 3. GC (Gas Chromatography) – Quality Control (Sr. Officer)
- 4. GLP & QMS Expert – Quality Control (Executive)
- 5. Production OSD – Officer (Manufacturing & Packing)
- 6. Machine Handling Operator – ALMEC 2500 (Production OSD)
- 7. Apprentice – Production Injection (Trainee)
- 8. Apprentice – Engineering (Trainee)
- Eligibility / Qualifications
- Location & Salary
- Application Process
- Why Consider a Pharma Career in Baddi?
- Frequently Asked Questions (FAQs)
- 1. Are freshers eligible?
- 2. Is prior pharma experience mandatory?
- 3. What instruments should QC candidates know?
- 4. Is this a walk-in interview?
- 5. What qualifications are accepted?
- Job Summary
BSc BPharm QC Production Jobs Baddi
B.Sc, M.Sc, B.Pharm vacancies in QC & Production at Adley Formulations, Baddi. Multiple openings for 0–8 yrs experience.
Adley Formulations Pvt. Ltd. is conducting a walk-in interview for multiple positions across Quality Control and Production departments at its formulation manufacturing facility in Baddi, Himachal Pradesh. Candidates with relevant experience in pharmaceutical OSD and injectable manufacturing are invited to attend. Freshers are also eligible for select apprenticeship roles.
If you are searching for pharma jobs in Baddi, QC HPLC analyst vacancies, analytical method validation careers, OSD production officer roles, or GLP QMS pharmaceutical jobs, this walk-in opportunity offers direct hiring access in a regulated manufacturing setup.
Company Overview
Adley Formulations Pvt. Ltd. operates in the pharmaceutical formulation manufacturing segment, serving domestic and regulated markets. Located in Baddi, one of India’s largest pharmaceutical hubs, the company focuses on quality-driven manufacturing aligned with GMP and regulatory compliance standards.
Pharmaceutical manufacturing in Baddi supports tablet, capsule, and injectable production for multiple therapeutic segments. Companies operating in this region maintain strict Quality Management Systems (QMS), Good Laboratory Practices (GLP), and audit-ready documentation processes to meet industry and regulatory requirements.
Working in such an environment offers professionals exposure to:
- USFDA and regulatory audit systems
- GMP-compliant production processes
- Advanced analytical instruments like HPLC and GC
- Analytical method validation and documentation control
- End-to-end formulation manufacturing
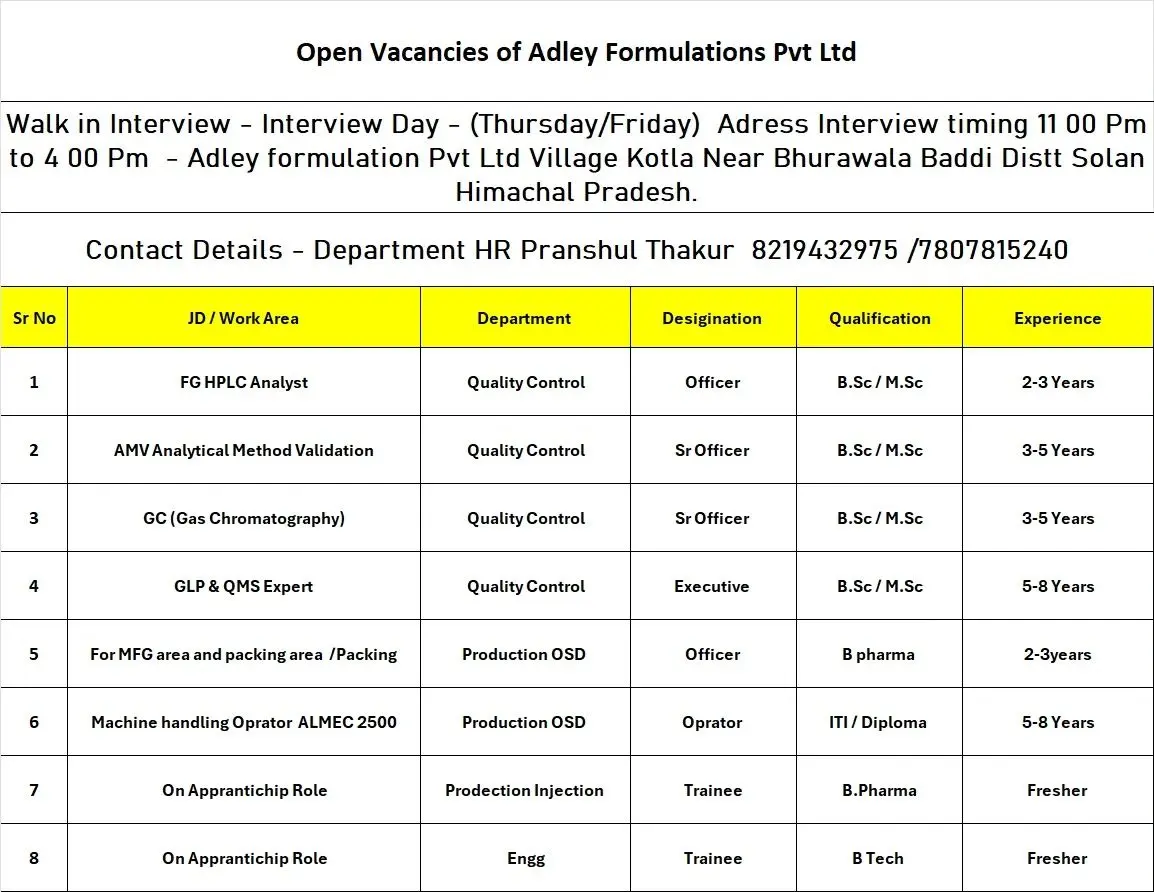
Walk-In Interview Details
Interview Days: Thursday / Friday
Interview Timings: 11:00 AM to 4:00 PM
Venue: Adley Formulations Pvt. Ltd., Village Kotla, Near Bhurawala, Baddi, District Solan, Himachal Pradesh
Contact (HR): Pranshul Thakur – 8219432975 / 7807815240
Candidates should carry updated resumes, educational documents, and relevant experience certificates.
Job Role & Responsibilities
1. FG HPLC Analyst – Quality Control (Officer)
Experience: 2–3 Years
Qualification: B.Sc / M.Sc
Key Responsibilities:
- Perform finished goods analysis using HPLC
- Execute routine QC testing as per approved specifications
- Maintain analytical documentation and calibration records
- Ensure compliance with GLP and GMP standards
- Support OOS and deviation investigations
This role is suitable for candidates with hands-on HPLC experience in a pharmaceutical QC laboratory.
2. AMV – Analytical Method Validation (Sr. Officer)
Experience: 3–5 Years
Qualification: B.Sc / M.Sc
Key Responsibilities:
- Conduct analytical method validation as per ICH guidelines
- Perform validation parameters including accuracy, precision, robustness, linearity
- Prepare validation protocols and reports
- Review raw data and ensure data integrity compliance
Strong understanding of analytical method development and regulatory documentation is essential.
3. GC (Gas Chromatography) – Quality Control (Sr. Officer)
Experience: 3–5 Years
Qualification: B.Sc / M.Sc
Key Responsibilities:
- Operate and maintain GC instruments
- Perform solvent residue and impurity analysis
- Maintain instrument qualification and calibration records
- Ensure compliance with QMS procedures
Candidates must have hands-on GC troubleshooting experience.
4. GLP & QMS Expert – Quality Control (Executive)
Experience: 5–8 Years
Qualification: B.Sc / M.Sc
Key Responsibilities:
- Implement and monitor GLP systems
- Manage QMS documentation and audit readiness
- Handle deviations, CAPA, and change control documentation
- Support internal and external audits
This is a high-responsibility role requiring strong knowledge of pharmaceutical quality systems and compliance frameworks.
5. Production OSD – Officer (Manufacturing & Packing)
Experience: 2–3 Years
Qualification: B.Pharm
Key Responsibilities:
- Supervise OSD manufacturing processes
- Oversee compression, granulation, coating, and packing activities
- Ensure adherence to batch manufacturing records (BMR)
- Monitor in-process checks and line clearance
Experience in tablet and capsule production lines is preferred.
6. Machine Handling Operator – ALMEC 2500 (Production OSD)
Experience: 5–8 Years
Qualification: ITI / Diploma
Key Responsibilities:
- Operate ALMEC 2500 machinery
- Perform preventive maintenance and troubleshooting
- Ensure compliance with SOPs and safety standards
Candidates must have prior exposure to pharmaceutical packing or production machinery.
7. Apprentice – Production Injection (Trainee)
Experience: Fresher
Qualification: B.Pharm
Key Responsibilities:
- Assist in injectable production activities
- Follow SOPs under supervision
- Support batch documentation and line activities
Fresh graduates looking for pharmaceutical manufacturing careers can apply.
8. Apprentice – Engineering (Trainee)
Experience: Fresher
Qualification: B.Tech
Key Responsibilities:
- Assist engineering maintenance team
- Support equipment monitoring and preventive maintenance
- Learn pharmaceutical plant engineering systems
This role provides entry-level exposure to pharmaceutical plant utilities and maintenance.
Eligibility / Qualifications
Relevant Courses (comma-separated):
B.Sc Chemistry, M.Sc Chemistry, B.Pharm, ITI, Diploma Engineering, B.Tech
Experience Range:
- Freshers for apprenticeship roles
- 2–8 years for experienced QC and Production roles
Industry Background Required:
Pharmaceutical formulation manufacturing, OSD plant, Injectable manufacturing, GMP-regulated environment
Location & Salary
Location: Baddi, District Solan, Himachal Pradesh
Industry Hub: Major pharmaceutical manufacturing cluster in North India
Salary will be offered as per industry standards and candidate experience. Senior QC and QMS roles typically command competitive compensation due to regulatory compliance expertise.
Application Process
Candidates can attend the walk-in interview directly on Thursday or Friday between 11:00 AM and 4:00 PM.
For queries, contact HR:
Pranshul Thakur
8219432975 / 7807815240
Candidates should bring:
- Updated resume
- Educational certificates
- Experience certificates
- ID proof
Why Consider a Pharma Career in Baddi?
Baddi is one of India’s largest pharmaceutical manufacturing zones. Professionals working here gain exposure to:
- GMP and GLP regulated environments
- Advanced analytical instruments like HPLC and GC
- Analytical method validation practices
- OSD and injectable manufacturing systems
- Quality assurance and audit compliance
QC analysts, QMS professionals, and production officers in regulated plants are in high demand due to increasing regulatory scrutiny and global export requirements.
Frequently Asked Questions (FAQs)
1. Are freshers eligible?
Yes, freshers can apply for apprenticeship roles in Production Injection and Engineering.
2. Is prior pharma experience mandatory?
Yes, for experienced QC, QMS, and Production roles, pharmaceutical background is required.
3. What instruments should QC candidates know?
HPLC and GC knowledge is essential for analytical roles.
4. Is this a walk-in interview?
Yes, candidates must attend the walk-in at the Baddi facility.
5. What qualifications are accepted?
B.Sc, M.Sc, B.Pharm, ITI, Diploma, and B.Tech depending on the role.
Job Summary
| Company | Adley Formulations Pvt. Ltd. |
|---|---|
| Vacancies | FG HPLC Analyst, AMV Sr Officer, GC Sr Officer, GLP & QMS Executive, Production OSD Officer, Machine Operator (ALMEC 2500), Apprentice – Injection, Apprentice – Engineering |
| Required Education | B.Sc, M.Sc, B.Pharm, ITI, Diploma, B.Tech |
| Experience | Fresher to 8 Years (Role Dependent) |