BDR Pharma Walk-In API QA QC R&D

- Company Overview
- Job Role & Responsibilities
- API Production (Oncology, General API, SPD)
- Analytical Development Lab (ADL – API)
- Regulatory Affairs (API)
- Engineering (Electrical, Mechanical, Instrumentation)
- Warehouse (API & OSD)
- Quality Assurance (API – Dispatch & IPQA)
- IT (CSV & Infrastructure)
- Microbiology QC (OSD)
- R&D (Peptide, Polymorphism, API Synthesis)
- Analytical Method Validation
- OSD Production (Granulation & Compression)
- Eligibility / Qualifications
- Location & Salary
- Application Process
- Why This Opportunity Stands Out
- FAQs
- Summary Table
BDR Pharma Walk-In API QA QC R&D Hiring 2026
BDR Pharma hiring 1–15 yrs candidates for multiple roles in Vadodara. ITI, BSc, MSc, BPharm eligible for API, QA, QC, R&D vacancies.
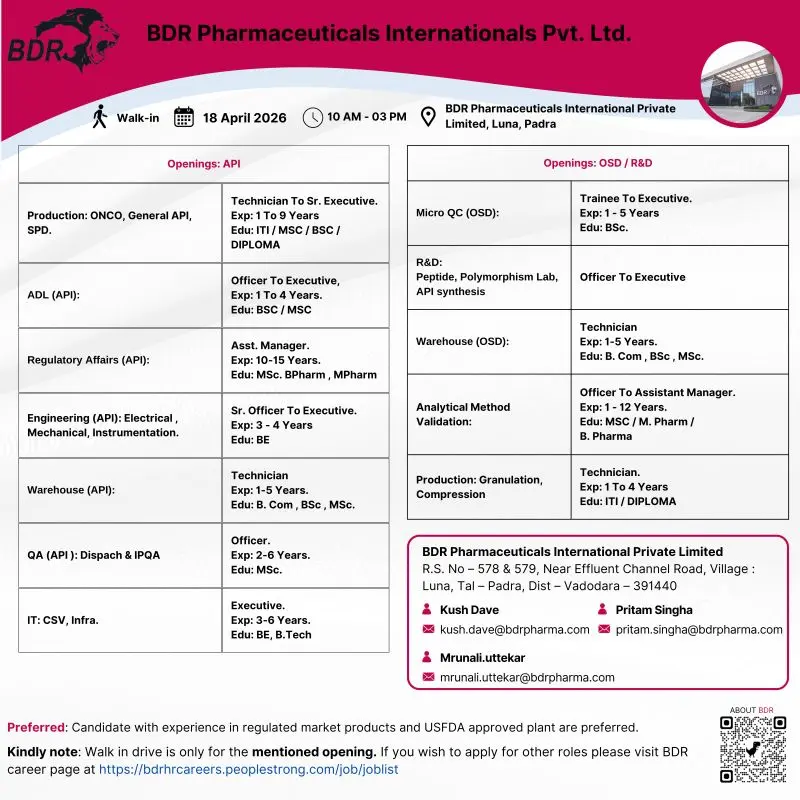
BDR Pharmaceuticals International Pvt. Ltd. is conducting a major walk-in drive for multiple vacancies across API Production, Quality Assurance, Quality Control, Regulatory Affairs, Engineering, R&D, and Warehouse departments. This hiring drive offers diverse job roles ranging from Technician to Senior Executive and Assistant Manager positions at their Luna, Padra (Vadodara) facility.
The company is hiring candidates with experience ranging from 1 to 15 years across pharmaceutical manufacturing, analytical development, regulatory affairs, and engineering domains. This is a strong opportunity for professionals looking to work in USFDA-approved pharma manufacturing environments and regulated market products.
Company Overview
BDR Pharmaceuticals International Pvt. Ltd. is a well-established pharmaceutical company known for its expertise in oncology products, API manufacturing, and finished dosage formulations. The company operates with global regulatory standards and has a strong presence in regulated markets, including USFDA-compliant facilities.
With a focus on innovation, high-quality drug development, and compliance-driven manufacturing, BDR Pharmaceuticals plays a key role in delivering affordable and effective medicines across therapeutic segments. The company provides excellent career growth opportunities in pharmaceutical production, research, and regulatory functions.
Job Role & Responsibilities
API Production (Oncology, General API, SPD)
- Execute batch manufacturing processes in API production units
- Handle equipment operations and ensure GMP compliance
- Maintain documentation and follow safety protocols
Analytical Development Lab (ADL – API)
- Perform analytical method development and validation
- Work on HPLC, GC, and other analytical instruments
- Support R&D and quality control functions
Regulatory Affairs (API)
- Prepare and review regulatory submissions
- Ensure compliance with global regulatory guidelines
- Coordinate with cross-functional teams for dossier preparation
Engineering (Electrical, Mechanical, Instrumentation)
- Maintain plant utilities and production equipment
- Troubleshoot and manage preventive maintenance
- Ensure smooth operations in manufacturing units
Warehouse (API & OSD)
- Manage material handling, storage, and inventory control
- Ensure compliance with GMP and documentation standards
Quality Assurance (API – Dispatch & IPQA)
- Conduct in-process quality checks and audits
- Review batch records and ensure compliance
- Handle deviations, CAPA, and documentation
IT (CSV & Infrastructure)
- Manage computer system validation and IT infrastructure
- Ensure data integrity and system compliance
Microbiology QC (OSD)
- Perform microbiological testing and environmental monitoring
- Maintain lab records and compliance standards
R&D (Peptide, Polymorphism, API Synthesis)
- Work on formulation and process development
- Support innovation in drug development
Analytical Method Validation
- Validate analytical methods as per regulatory standards
- Prepare validation protocols and reports
OSD Production (Granulation & Compression)
- Operate manufacturing equipment for solid dosage forms
- Maintain production efficiency and quality standards
Eligibility / Qualifications
Candidates from the following educational backgrounds can apply:
ITI, Diploma, BSc, MSc, B.Pharm, M.Pharm, B.Com, BE, B.Tech
Relevant courses include: Pharmacy, Pharmaceutical Chemistry, Analytical Chemistry, Microbiology, Biotechnology, Mechanical Engineering, Electrical Engineering, Instrumentation, Regulatory Affairs
- Experience required: 1 to 15 years depending on role
- Experience in USFDA-approved plants and regulated markets preferred
- Strong knowledge of GMP, GLP, and pharma manufacturing processes is an advantage
Location & Salary
- Walk-in Location: BDR Pharmaceuticals International Pvt. Ltd., Luna, Padra, Vadodara
- Date: 18 April 2026
- Time: 10:00 AM to 03:00 PM
- Salary: As per industry standards based on experience
Application Process
Candidates can attend the walk-in interview directly at the venue or share their resume via email:
Email Contacts:
For other roles, candidates can apply through the official career page:
https://bdrhrcareers.peoplestrong.com/job/joblist
Why This Opportunity Stands Out
- Opportunity to work in USFDA-approved pharmaceutical facilities
- Exposure to oncology API manufacturing and advanced R&D
- Multiple openings across technical and non-technical pharma domains
- Strong career growth in regulated pharma markets
FAQs
Who can attend this walk-in drive?
Candidates with relevant pharma, science, and engineering qualifications and required experience can attend.
Is experience in regulated markets required?
Preferred but not mandatory for all roles.
What roles are available in production?
API and OSD production roles including granulation and compression.
Can freshers apply?
Most roles require experience; freshers may have limited opportunities.
How to apply if unable to attend walk-in?
You can send your CV to the provided email addresses or apply via the career portal.
Summary Table
| Category | Details |
|---|---|
| Company | BDR Pharmaceuticals International Pvt. Ltd. |
| Vacancies | Production Executive, QA Officer, QC Analyst, ADL Executive, Regulatory Affairs Executive, Engineering Manager, Warehouse Technician, IT Executive, R&D Executive |
| Required Education | ITI, Diploma, BSc, MSc, B.Pharm, M.Pharm, B.Com, BE, B.Tech |
| Experience | 1 to 15 Years |
You must sign in to apply for this position.


