Macleods walk-in API, OSD, QC, DPI & Engineering

- Company Overview
- Job Role & Responsibilities
- Production (API)
- Production (OSD)
- Engineering Services
- Quality Control (QC – OSD)
- Production (DPI)
- Eligibility / Qualifications
- Educational Qualification
- Relevant Courses
- Experience Required
- Preferred Technical Skills
- Why Pharmaceutical Manufacturing Careers Offer Long-Term Growth
- Work Environment & Career Growth
- Location & Salary
- Job Location
- Approximate Salary
- Application Process
- Registration Link
- Walk-In Interview Date
- Interview Timing
- Walk-In Venue
- Documents Required
- Contact Numbers
- Important Note
- FAQs
- 1. Which company is conducting this pharma walk-in drive?
- 2. Which departments are hiring?
- 3. What qualifications are eligible for Macleods jobs?
- 4. What is the experience requirement?
- 5. What is the interview location?
- 6. What technical skills are preferred?
- Summary Table
Meta Title: Macleods Pharma Jobs | QC Production | Vapi
Meta Description: Macleods hiring for API, OSD, QC, DPI & Engineering roles in Vapi, Daman & Sarigam. BSc, MSc, BPharm eligible.
Macleods Pharmaceuticals has announced a major walk-in interview drive at Vapi for multiple openings across API Production, OSD Production, Engineering Services, Quality Control, and DPI manufacturing departments. Candidates looking for pharmaceutical manufacturing jobs in Gujarat, API production careers, QC analyst jobs, formulation manufacturing roles, engineering pharma jobs, and walk-in pharma interviews can explore this hiring opportunity.
The recruitment drive is open for experienced pharmaceutical professionals with backgrounds in API manufacturing, formulation production, engineering maintenance, analytical quality control, and sterile manufacturing operations.
Candidates with qualifications such as B.Sc, M.Sc Chemistry, B.Pharm, M.Pharm, B.E, B.Tech, ITI, Diploma, and graduation backgrounds are eligible for various technical and operational roles.
This latest pharma walk-in drive is expected to attract candidates searching for regulated pharmaceutical manufacturing jobs, GMP pharma careers, OSD production jobs, HVAC maintenance careers, and analytical laboratory opportunities.
Company Overview
Macleods Pharmaceuticals is one of India’s leading pharmaceutical companies with a strong presence in regulated pharmaceutical manufacturing, API production, oral solid dosage manufacturing, and global pharmaceutical exports.
The company operates advanced pharmaceutical facilities focused on:
- API manufacturing operations
- OSD production systems
- DPI manufacturing
- Engineering utility management
- Pharmaceutical quality control
- HVAC and water system operations
- Sterile manufacturing technologies
- Pharmaceutical compliance systems
Macleods has established itself as a major player in regulated pharmaceutical manufacturing through strong quality systems, global compliance standards, and advanced manufacturing infrastructure.
The company follows strict compliance with:
- GMP regulations
- GLP guidelines
- Pharmaceutical quality systems
- Regulatory documentation standards
- Manufacturing SOPs
- Analytical quality procedures
- Validation systems
- Utility compliance standards
Professionals working at Macleods gain exposure to modern pharmaceutical manufacturing technologies and regulated production environments.
Job Role & Responsibilities
Macleods Pharmaceuticals is hiring candidates for API Production, OSD Production, Engineering Services, Quality Control, and DPI Production departments.
Selected candidates will work in GMP-regulated pharmaceutical environments involving production operations, analytical testing, engineering systems, and manufacturing support activities.
Production (API)
API Production professionals will support active pharmaceutical ingredient manufacturing and intermediate production operations.
Key Responsibilities
- Handling API manufacturing operations
- Supporting SRP manufacturing activities
- Monitoring chemical process operations
- Executing shift production activities
- Maintaining batch manufacturing records
- Following SOP and GMP documentation practices
- Operating manufacturing equipment
- Handling intermediates manufacturing systems
- Production troubleshooting and process monitoring
Candidates with API manufacturing and intermediate production experience may receive preference.
Production (OSD)
OSD Production candidates will support formulation manufacturing and packing operations.
Key Responsibilities
- Handling bulk packing operations
- Managing primary and secondary packing activities
- Supporting formulation production systems
- Operating packaging machines
- Maintaining production records
- Ensuring GMP compliance during manufacturing
- Monitoring shift production activities
- Supporting packing line operations
Candidates with oral solid dosage manufacturing exposure are encouraged to attend the walk-in drive.
Engineering Services
Engineering professionals will support pharmaceutical utility systems and plant maintenance operations.
Key Responsibilities
- Managing plant maintenance activities
- HVAC system monitoring
- Utility system maintenance
- Water system operations
- Instrumentation support
- Electrical and mechanical maintenance
- Preventive maintenance activities
- Utility troubleshooting and repair
- Engineering compliance documentation
Candidates with formulation plant engineering exposure may receive preference.
Quality Control (QC – OSD)
Quality Control professionals will support analytical laboratory operations and pharmaceutical testing systems.
Key Responsibilities
- Analytical validation activities
- Finished goods testing
- Raw material analysis
- Stability testing activities
- Handling GLP compliance systems
- Analytical documentation review
- Instrument operation and calibration
- Laboratory investigation support
- Maintaining GMP laboratory standards
Candidates with exposure to analytical validation and pharmaceutical QC operations are encouraged to apply.
Production (DPI)
DPI Production candidates will support sterile manufacturing and filling operations.
Key Responsibilities
- Operating vial filling and sealing machines
- CRAB system operations
- Autoclave handling
- Tunnel operations
- Vial washing activities
- IPC and media fill support
- Sterile manufacturing operations
- Production documentation management
- GMP compliance activities
Candidates with sterile manufacturing and injectable production experience may receive preference.
Eligibility / Qualifications
Candidates applying for Macleods pharmaceutical jobs should satisfy the eligibility criteria mentioned below.
Educational Qualification
API Production
- B.Sc Chemistry
- M.Sc Chemistry
- B.E Chemical
- B.Tech Chemical
- Diploma Chemical
OSD Production
- ITI
- Diploma
- Any Graduate
Engineering Services
- B.E Electrical
- B.Tech Electrical
- B.E Mechanical
- B.Tech Mechanical
- ITI
- Diploma
Quality Control
- B.Pharm
- M.Pharm
- M.Sc Chemistry
DPI Production
- ITI
- Diploma
- Any Graduate
Relevant Courses
Industrial Chemistry, Organic Chemistry, Analytical Chemistry, Pharmaceutical Technology, Pharmaceutical Analysis, Industrial Pharmacy, Mechanical Engineering, Electrical Engineering, Chemical Engineering, Instrumentation Technology, Pharmaceutical Manufacturing Technology, HVAC Systems, Utility Management
Experience Required
API Production
- 2 to 8 Years
OSD Production
- 2 to 8 Years
Engineering Services
- 2 to 8 Years
Quality Control
- 2 to 6 Years
DPI Production
- 2 to 6 Years
Preferred Technical Skills
Candidates with the following pharmaceutical skills may receive preference:
- API manufacturing
- OSD manufacturing
- HVAC systems
- Utility maintenance
- Water systems
- Analytical validation
- Stability testing
- GMP documentation
- GLP compliance
- Sterile manufacturing
- Vial filling operations
- CRAB systems
- Media fill activities
- Primary and secondary packing
- Engineering maintenance
Why Pharmaceutical Manufacturing Careers Offer Long-Term Growth
India’s pharmaceutical manufacturing sector continues to generate strong demand for experienced professionals across:
- API manufacturing
- Formulation production
- Engineering maintenance
- Quality control laboratories
- Sterile manufacturing operations
- Pharmaceutical packaging
- Utility and HVAC systems
- Regulatory compliance operations
Professionals with regulated manufacturing exposure often receive excellent career growth opportunities due to increasing exports and expansion of pharmaceutical production capacities.
Candidates working in companies like Macleods gain experience in:
- Global manufacturing systems
- Regulated pharmaceutical operations
- Analytical quality systems
- Utility engineering management
- Sterile production technologies
- Pharmaceutical process optimization
- Advanced manufacturing practices
Work Environment & Career Growth
Macleods Pharmaceuticals offers employees exposure to highly regulated pharmaceutical operations and advanced manufacturing systems.
Professionals joining the organization may gain hands-on experience in:
- API manufacturing operations
- OSD formulation systems
- Engineering and utility management
- HVAC and water system maintenance
- Analytical quality testing
- Pharmaceutical validation systems
- Production documentation practices
- Sterile manufacturing technologies
Employees with strong technical and compliance expertise may grow into senior production, engineering, quality, and operational leadership roles.
Location & Salary
Job Location
Vapi / Daman / Sarigam
Approximate Salary
Based on current pharmaceutical industry salary trends in Gujarat regulated manufacturing plants, the approximate salary may range between ₹2.8 LPA to ₹10 LPA depending on role, experience, technical skills, and department.
Application Process
Interested candidates should register through the official registration link before attending the walk-in interview.
Registration Link
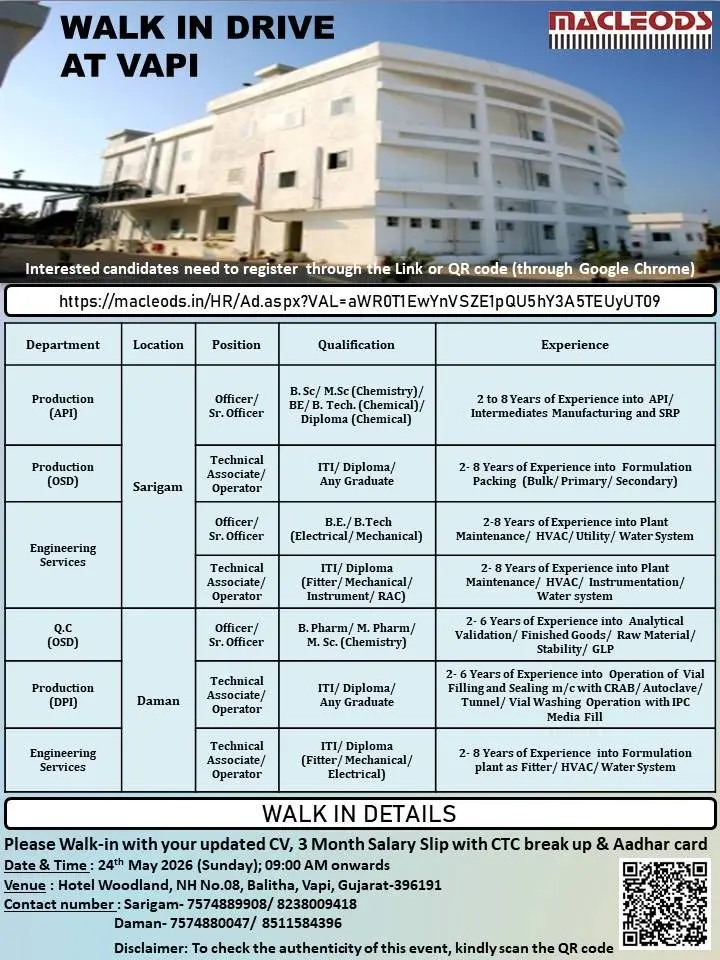
https://macleods.in/HR/Ad.aspx?VAL=aWROT1EwYnVSZE1pQU5hY3A5TEUYUT09
Walk-In Interview Date
24th May 2026 (Sunday)
Interview Timing
09:00 AM onwards
Walk-In Venue
Hotel Woodland, NH No.08, Balitha, Vapi, Gujarat – 396191
Documents Required
Candidates should carry:
- Updated CV
- Last 3 months salary slips
- CTC breakup
- Aadhar card
- Educational certificates
- Experience documents
Contact Numbers
Sarigam
- 7574889908
- 8238009418
Daman
- 7574880047
- 8511584396
Important Note
Candidates are advised to complete registration through the official link before attending the interview.
Experienced professionals with regulated pharmaceutical manufacturing exposure and GMP knowledge are strongly encouraged to apply.
FAQs
1. Which company is conducting this pharma walk-in drive?
Macleods Pharmaceuticals is conducting the walk-in interview drive.
2. Which departments are hiring?
API Production, OSD Production, Engineering Services, QC, and DPI Production departments are hiring.
3. What qualifications are eligible for Macleods jobs?
B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma, B.E, and B.Tech candidates are eligible depending on the role.
4. What is the experience requirement?
Candidates with 2 to 8 years of pharmaceutical manufacturing experience can apply.
5. What is the interview location?
The walk-in interview venue is Hotel Woodland, Vapi, Gujarat.
6. What technical skills are preferred?
API manufacturing, analytical validation, HVAC systems, vial filling, sterile manufacturing, GLP compliance, and utility maintenance skills are preferred.
Summary Table
| Company | Macleods Pharmaceuticals |
| Department Vacancies | API Production, OSD Production, QC, DPI, Engineering |
| Qualification | B.Sc, M.Sc, B.Pharm, M.Pharm, ITI, Diploma, B.E, B.Tech |
| Experience | 2 to 8 Years |
| Location | Vapi, Daman, Sarigam |
To apply for this job please visit macleods.in.


