PharmaZone walk-in BD, CRA, GMP, Production, PV, MICC, RA

- Company Overview
- Job Role & Responsibilities
- Business Development – Executive
- Clinical Research Associate (CRA) – GCP Services
- GMP Auditor – GMP Services
- Production Expert – OSD
- Pharmacovigilance – Senior Executive (Aggregate Reports)
- MICC – Medical Information Call Centre
- Regulatory Affairs – Senior Executive
- Eligibility / Qualifications
- Location & Walk-in Details
- Application Process
- FAQs
- Summary Table
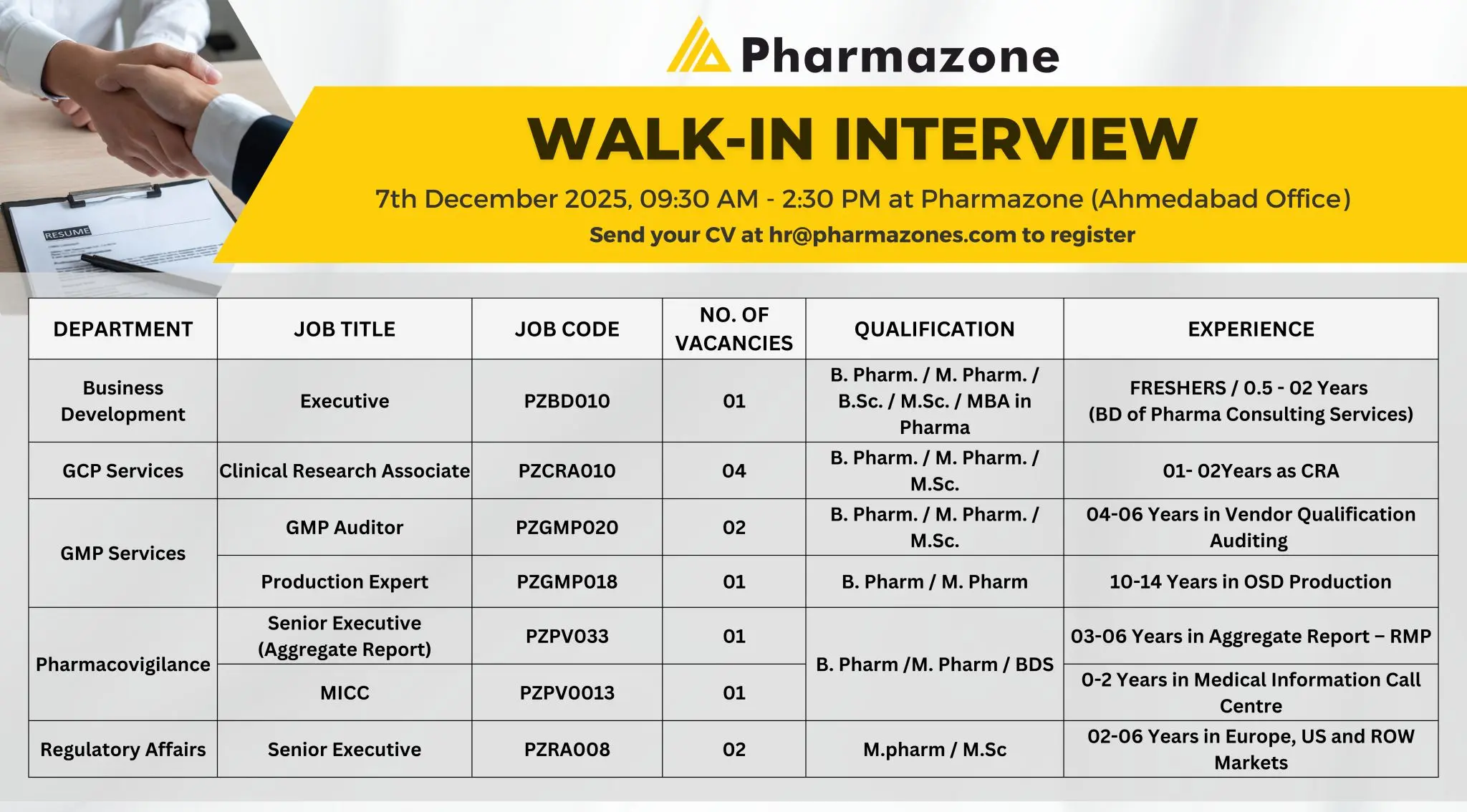
B.Pharm/M.Pharm Openings – 13 Vacancies – Ahmedabad
Apply for 13 vacancies at PharmaZone Ahmedabad. Openings in BD, GMP, CRA, PV, MICC & RA for B.Pharm/M.Pharm/B.Sc/M.Sc candidates.
PharmaZone is conducting a walk-in interview for multiple clinical research, GMP auditing, pharmacovigilance, production, and regulatory affairs roles. These positions suit candidates looking to advance their careers in high-growth pharma consulting and global compliance services.
Company Overview
PharmaZone is a well-established pharmaceutical consulting and services organization supporting global clients across GCP, GMP, regulatory affairs, pharmacovigilance, and medical information. With strong domain expertise and compliance-driven workflows, the company provides end‑to‑end support for drug development, audits, regulatory submissions, and safety operations.
Job Role & Responsibilities
Business Development – Executive
- Support BD activities for pharma consulting services.
- Coordinate with clients for proposals and service offerings.
- Assist in outreach, lead generation, and follow-ups.
Clinical Research Associate (CRA) – GCP Services
- Perform site monitoring and documentation as per GCP.
- Support investigator meetings, compliance checks, and study execution.
GMP Auditor – GMP Services
- Conduct vendor qualification audits.
- Prepare audit reports and track CAPA.
- Ensure compliance with global GMP regulations.
Production Expert – OSD
- Supervise OSD manufacturing operations.
- Review BMR/BPR and oversee production compliance.
Pharmacovigilance – Senior Executive (Aggregate Reports)
- Prepare PSURs, PBRERs, RMPs.
- Perform literature screening and safety data analysis.
MICC – Medical Information Call Centre
- Handle customer queries regarding product safety and usage.
- Maintain accurate call documentation.
Regulatory Affairs – Senior Executive
- Manage EU, US & ROW submissions.
- Compile dossiers and respond to regulatory queries.
Eligibility / Qualifications
- BD Executive: B.Pharm, M.Pharm, B.Sc, M.Sc, MBA – Freshers to 2 years.
- CRA: B.Pharm, M.Pharm, M.Sc – 1–2 years.
- GMP Auditor: B.Pharm, M.Pharm, M.Sc – 4–6 years.
- Production Expert: B.Pharm, M.Pharm – 10–14 years.
- PV Senior Executive: B.Pharm, M.Pharm, BDS – 3–6 years.
- MICC: Any Life Science Graduate – 0–2 years.
- RA Senior Executive: M.Pharm, M.Sc – 2–6 years.
Location & Walk-in Details
Walk-In Date: 7th December 2025
Time: 09:30 AM – 2:30 PM
Venue: PharmaZone, Ahmedabad Office.
Application Process
To register for the walk‑in, email your CV to: hr@pharmazones.com
FAQs
1. Who can attend the walk-in?
Life science graduates with the specified experience for each role.
2. Is pharma consulting experience required?
Required only for specific BD and specialist roles.
3. Are freshers eligible?
Yes, BD and MICC roles accept freshers.
4. Are these office-based roles?
Yes, roles are based at the Ahmedabad office unless stated otherwise.
5. How many vacancies are available?
A total of 13 positions across multiple departments.
Summary Table
| Category | Details |
|---|---|
| Company | PharmaZone |
| Vacancies | 13 total across BD, CRA, GMP, Production, PV, MICC, RA |
| Required Education | B.Pharm, M.Pharm, B.Sc, M.Sc, MBA, BDS |
| Experience | Freshers to 14 years depending on role |