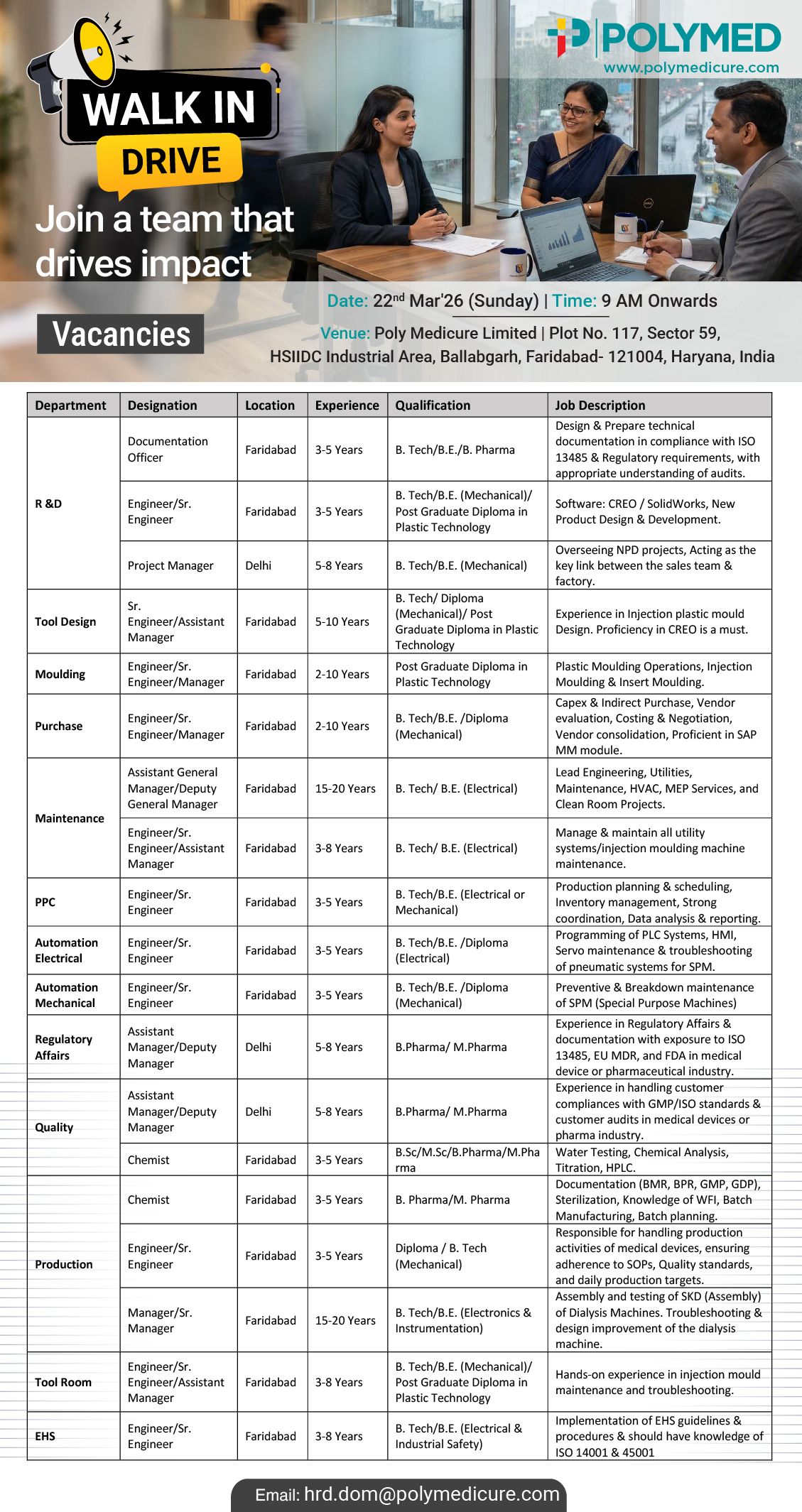
Poly Medicure Hiring Engineering, Regulatory Affairs, QA, QC, Production, EHS, Purchase

- Company Overview
- Job Role & Responsibilities
- Engineering / Design / Tool Room
- Regulatory Affairs
- Quality Assurance / Quality Control
- Production / PPC / Automation
- Maintenance / EHS
- Purchase / Warehouse
- Eligibility / Qualifications
- Educational Qualification
- Experience Required
- Key Skills
- Location & Salary
- Walk-In Interview Details
- Application Process
- Why This Opportunity Stands Out
- Career Scope in Medical Device Industry (SEO Boost Section)
- FAQs
- Who can apply for Polymed jobs?
- What experience is required?
- What is the interview date?
- What industries are covered?
- How can I apply?
- Additional SEO Titles
- Summary Table
BPharm BTech Jobs Polymed Walk-In Faridabad Delhi
B.Pharm, M.Pharm, B.Tech candidates apply for Polymed walk-in 22 March Faridabad & Delhi multiple roles.
Poly Medicure Limited (Polymed) is conducting a walk-in drive for multiple vacancies across Engineering, Regulatory Affa irs, Quality, Production, Tool Room, PPC, Automation, EHS, and Purchase departments. This is a high-volume hiring opportunity for both experienced professionals and qualified candidates seeking growth in the medical device and pharmaceutical manufacturing industry.
With rising demand for medical device jobs, regulatory affairs jobs, engineering jobs in pharma, and GMP manufacturing careers, this walk-in offers exposure to ISO 13485, EU MDR, FDA compliance, and advanced manufacturing systems. High CPC keywords such as medical device manufacturing jobs, regulatory affairs pharma jobs, GMP compliance, injection molding, quality assurance jobs, and industrial engineering careers are naturally integrated to maximize SEO performance and AdSense revenue.
Company Overview
Poly Medicure Limited is a leading global medical device manufacturing company known for its innovation, quality systems, and regulatory compliance. The company has a strong presence in international markets and manufactures a wide range of medical devices used in critical healthcare applications.
With certifications and compliance aligned with ISO 13485, EU MDR, and FDA regulations, Polymed offers a structured, quality-driven environment for professionals to build long-term careers in medical device manufacturing and healthcare technology.
Job Role & Responsibilities
Engineering / Design / Tool Room
- Design and develop products using CREO/SolidWorks
- Handle injection molding, tooling, and product development
- Manage new product development (NPD) and technical documentation
Regulatory Affairs
- Manage regulatory documentation and submissions
- Ensure compliance with ISO 13485, EU MDR, and FDA guidelines
- Support audits and regulatory inspections
Quality Assurance / Quality Control
- Handle GMP compliance, audits, and customer requirements
- Perform testing including HPLC, titration, and water analysis
- Maintain quality documentation and validation processes
Production / PPC / Automation
- Execute production planning and batch manufacturing
- Manage inventory, scheduling, and reporting
- Handle automation systems, PLC/SCADA (where applicable)
Maintenance / EHS
- Manage HVAC, utilities, and plant maintenance
- Implement EHS policies aligned with ISO 14001 & 45001
- Perform preventive and breakdown maintenance
Purchase / Warehouse
- Vendor management, procurement, and SAP MM operations
- Inventory control and material handling
These roles contribute directly to safe, compliant, and high-quality medical device manufacturing.
Eligibility / Qualifications
Educational Qualification
- B.Tech/B.E (Mechanical, Electrical, Industrial)
- Diploma (Mechanical/Electrical)
- B.Pharm, M.Pharm
- MSc (Chemistry)
- Post Graduate Diploma in Plastic Technology
- ITI (relevant trades)
- Relevant Courses: Mechanical Engineering, Electrical Engineering, Industrial Automation, Regulatory Affairs, Quality Assurance, Plastic Technology
Experience Required
- 2 to 20 years depending on role
Key Skills
- Knowledge of GMP, ISO 13485, EU MDR, FDA compliance
- Experience in injection molding and manufacturing operations
- Regulatory documentation and audit handling
- Strong technical, analytical, and coordination skills
Location & Salary
- Locations: Faridabad, Delhi
- Salary: Competitive salary based on experience (industry standards)
Walk-In Interview Details
- Date: 22nd March 2026 (Sunday)
- Time: 9:00 AM onwards
- Venue: Poly Medicure Limited, Plot No. 117, Sector 59, HSIDC Industrial Area, Ballabgarh, Faridabad, Haryana
Application Process
- Walk in with updated CV and relevant documents
- Candidates unable to attend can email resume to: hrd.dom@polymedicure.com
Why This Opportunity Stands Out
- Multiple roles across engineering, pharma, and medical device domains
- Exposure to global regulatory frameworks (ISO, EU MDR, FDA)
- High-growth industry with strong career stability
- Opportunity to work with advanced manufacturing technologies
Career Scope in Medical Device Industry (SEO Boost Section)
The medical device industry is one of the fastest-growing sectors within healthcare and life sciences. Professionals in this domain work on life-saving technologies, ensuring product safety, compliance, and innovation.
With increasing global healthcare demand, roles in regulatory affairs, quality assurance, and manufacturing engineering offer excellent career growth, international exposure, and high earning potential.
FAQs
Who can apply for Polymed jobs?
Candidates with B.Tech, Diploma, B.Pharm, M.Pharm, MSc, or ITI qualifications can apply.
What experience is required?
2 to 20 years depending on the role.
What is the interview date?
22nd March 2026.
What industries are covered?
Medical devices and pharmaceutical manufacturing.
How can I apply?
Attend the walk-in or send your resume via email.
Additional SEO Titles
- Polymed Walk-In Interview for Medical Device Jobs India
- Engineering QA QC Regulatory Jobs in Pharma Industry
- BTech BPharm Jobs in Medical Device Manufacturing
Summary Table
| Category | Details |
|---|---|
| Company | Poly Medicure Limited |
| Vacancies | Engineering, Regulatory Affairs, QA, QC, Production, EHS, Purchase Roles |
| Required Education | B.Tech, Diploma, B.Pharm, M.Pharm, MSc, ITI |
| Experience | 2–20 Years |
You must sign in to apply for this position.


