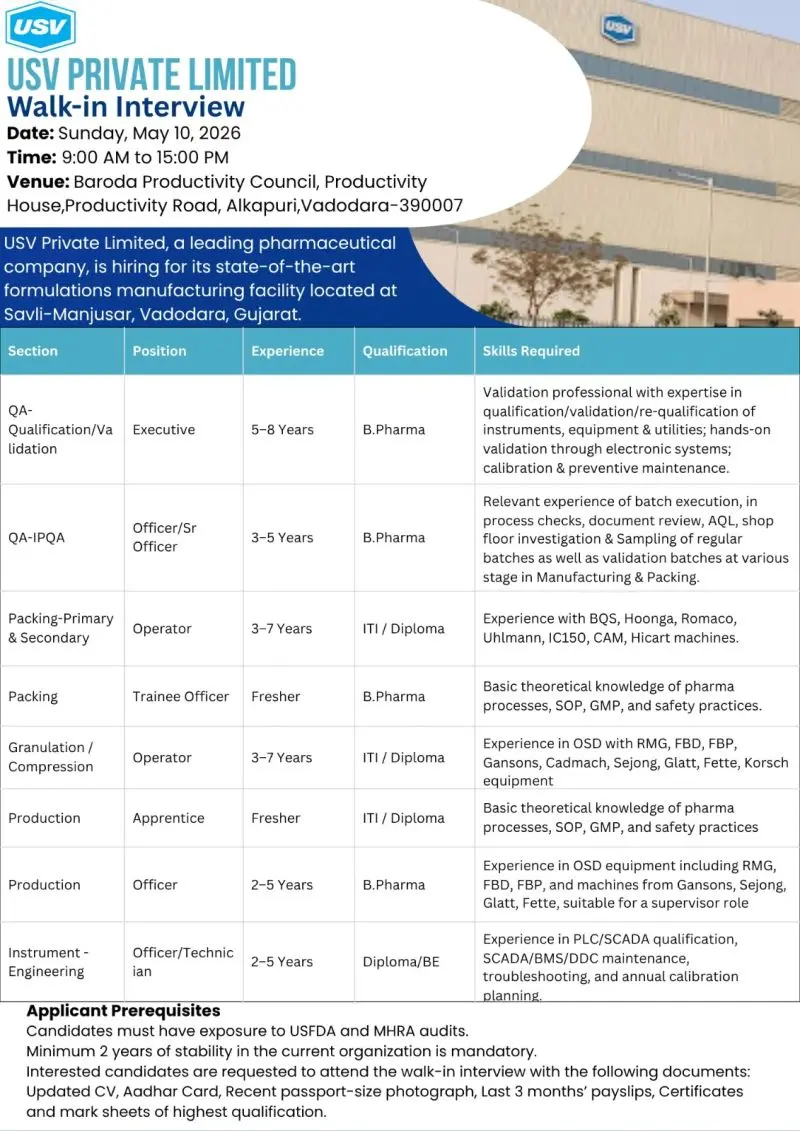
USV walk-in QA, QA, Packing,Engineering

- Company Overview
- Job Role & Responsibilities
- Quality Assurance (QA – Qualification/Validation & IPQA)
- Production (OSD – Granulation/Compression/Coating)
- Packing (Primary & Secondary)
- Engineering / Instrumentation
- Eligibility / Qualifications
- Educational Qualification:
- Experience Required:
- Skills Required:
- Location & Salary
- Interview Location:
- Job Location:
- Salary:
- Application Process
- Documents Required:
- Important Notes:
- Why Join USV?
- High CPC Keywords Integrated
- Frequently Asked Questions (FAQs)
- Who can apply for these USV roles?
- Is OSD experience mandatory?
- What audit exposure is expected?
- What is the selection process?
- What salary can I expect?
- Summary Table
BPharm ITI Pharma Walk-In 8+ Roles USV Vadodara
B.Pharm, ITI, Diploma candidates for 8+ roles at USV Vadodara. Fresher–8 yrs exp. Walk-in on May 10.
USV Private Limited is hosting a walk-in interview for multiple openings across Quality Assurance (QA), Production, Packing, and Engineering at its formulations facility in Savli–Manjusar, Vadodara. This drive targets both freshers and experienced professionals (0–8 years) with exposure to OSD manufacturing and regulated environments. If you’re aiming for pharma QA jobs, OSD production roles, or GMP-compliant manufacturing careers, this is a direct entry into a well-established, audit-ready organization.
Company Overview
USV Private Limited is a reputed pharmaceutical manufacturer with a strong presence in domestic and international markets. The company operates modern formulation facilities aligned with global regulatory standards including USFDA and MHRA. Known for its quality systems, documentation rigor, and process control, USV delivers reliable medicines across key therapeutic areas.
Its Savli–Manjusar site is equipped for Oral Solid Dosage (OSD) manufacturing with validated equipment, electronic systems, and audit-ready processes. Teams work within strict cGMP frameworks, ensuring data integrity, traceability, and consistent product quality.
Job Role & Responsibilities
Quality Assurance (QA – Qualification/Validation & IPQA)
- Execute equipment, utility, and instrument qualification/validation/re-qualification
- Perform in-process quality assurance (IPQA) checks during manufacturing and packing
- Review batch documents, sampling records, AQL results, and deviation investigations
- Support calibration, preventive maintenance, and electronic validation systems
- Ensure strict adherence to cGMP, SOPs, and data integrity principles
Production (OSD – Granulation/Compression/Coating)
- Operate and monitor OSD equipment such as RMG, FBD/FBP, granulators, compression and coating machines
- Execute batch manufacturing steps with proper documentation and line clearance
- Maintain process parameters, handle alarms, and ensure product quality at each stage
- Follow safety protocols and GMP practices on the shop floor
Packing (Primary & Secondary)
- Operate packaging lines (Uhlmann, CAM, Hicart, IC150, Romaco, Hoonga)
- Perform in-process checks, AQL inspections, and packaging verification
- Maintain batch packing records and ensure traceability and compliance
Engineering / Instrumentation
- Handle PLC/SCADA systems, BMS/DDC maintenance, and troubleshooting
- Execute annual calibration plans and preventive maintenance schedules
- Support qualification activities and ensure equipment compliance
These roles directly impact batch consistency, regulatory readiness, and overall manufacturing efficiency in a high-compliance environment.
Eligibility / Qualifications
Educational Qualification:
B.Pharm, ITI, Diploma, BE/B.Tech (Electrical, Mechanical, Instrumentation)
Relevant courses: Pharmaceutics, Industrial Pharmacy, Pharmaceutical Chemistry, Mechanical Engineering, Electrical Engineering, Instrumentation & Control, Electronics
Experience Required:
- QA Executive (Qualification/Validation): 5–8 years
- QA-IPQA Officer/Sr. Officer: 3–5 years
- Production Officer: 2–5 years
- Packing/Production Operators: 3–7 years
- Engineering Officer/Technician: 2–5 years
- Trainee Officer / Apprentice: Freshers (B.Pharm / ITI / Diploma)
Skills Required:
- Strong understanding of cGMP, SOPs, and safety practices
- Hands-on exposure to OSD equipment (RMG, FBD, FBP; Gansons, Cadmach, Sejong, Glatt, Fette, Korsch)
- Experience with packaging lines (Uhlmann, CAM, Hicart) and AQL practices
- Validation expertise: qualification, calibration, preventive maintenance, electronic systems
- Exposure to USFDA/MHRA audits and documentation expectations
Location & Salary
Interview Location:
Baroda Productivity Council, Productivity House, Productivity Road, Alkapuri, Vadodara – 390007
Job Location:
Savli–Manjusar, Vadodara, Gujarat
Salary:
As per industry standards (role- and experience-based; competitive packages for validated OSD/QA profiles)
Application Process
Attend the walk-in interview with required documents:
- Date: Sunday, 10 May 2026
- Time: 09:00 AM to 03:00 PM
- Venue: Baroda Productivity Council, Alkapuri, Vadodara
Documents Required:
- Updated CV
- Aadhar Card
- Recent passport-size photograph
- Last 3 months’ payslips
- Educational certificates and mark sheets
Important Notes:
- Minimum 2 years of stability in the current organization is required for experienced roles
- Candidates with USFDA/MHRA audit exposure will be prioritized
Why Join USV?
- Work in USFDA/MHRA-compliant facilities with audit-ready systems
- Exposure to advanced OSD manufacturing and validated equipment
- Structured growth across QA, Production, and Engineering tracks
- Strong emphasis on GMP, data integrity, and process excellence
High CPC Keywords Integrated
pharma walk-in interview Vadodara, USFDA pharma jobs India, QA validation jobs pharma, OSD production jobs India, GMP manufacturing jobs, HPLC/analytical exposure pharma, packing operator pharma jobs, pharma engineering jobs India, high paying pharma jobs India, regulatory compliance pharma careers
Frequently Asked Questions (FAQs)
Who can apply for these USV roles?
Candidates with B.Pharm, ITI, Diploma, or Engineering degrees with relevant experience can apply. Freshers can apply for trainee/apprentice roles.
Is OSD experience mandatory?
For operator and officer roles, OSD exposure (RMG, FBD, compression/coating) is preferred and often required.
What audit exposure is expected?
Experience with USFDA/MHRA audits and documentation practices is preferred.
What is the selection process?
Direct walk-in screening with technical evaluation and HR discussion. Carry all required documents.
What salary can I expect?
Compensation is competitive and aligned with experience, role, and audit exposure.
Summary Table
| Company | USV Private Limited |
|---|---|
| Vacancies | QA Executive (Qualification/Validation), QA-IPQA Officer/Sr. Officer, Production Officer/Operator, Packing Operator, Engineering Officer/Technician |
| Required Education | B.Pharm, ITI, Diploma, BE/B.Tech |
| Experience | Freshers to 8 Years |