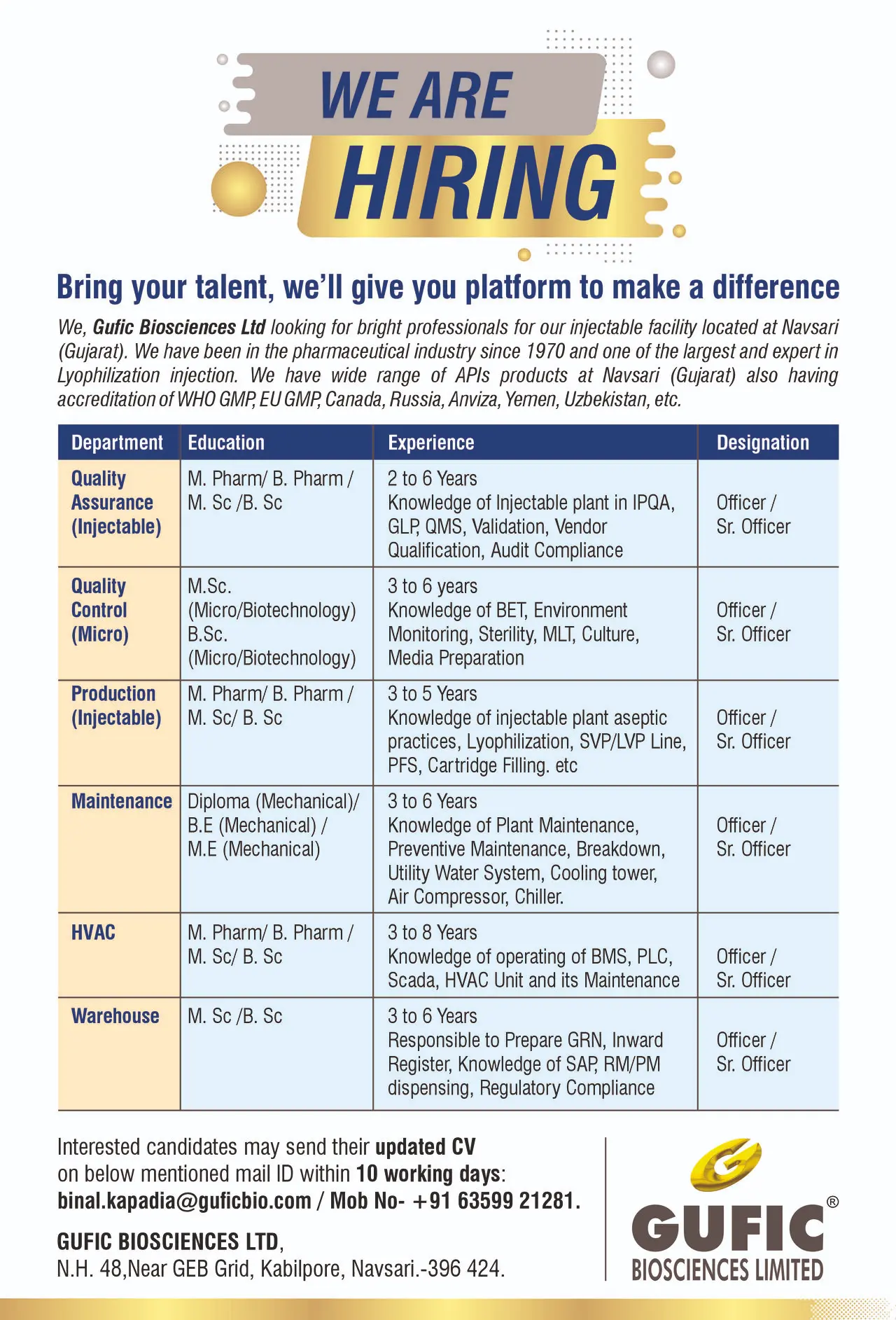
Gufic Hiring QA, QC, Production

- Company Overview
- Job Role & Responsibilities
- Quality Assurance (Injectables – IPQA & Compliance)
- Quality Control (Microbiology)
- Production (Injectables – Aseptic Operations)
- Maintenance (Engineering)
- HVAC (Engineering Systems)
- Warehouse (Materials Management)
- Eligibility / Qualifications
- Location & Salary
- Application Process
- Why This Opportunity Stands Out
- FAQs
- Summary Table
BPharm MSc Pharma Jobs Gufic Navsari Hiring
Gufic Biosciences hiring QA, QC, Production roles in Navsari. BPharm, MSc with 2–8 yrs experience. Multiple vacancies open.
Gufic Biosciences Ltd is actively hiring for multiple vacancies across Quality Assurance, Quality Control (Microbiology), Production (Injectables), Maintenance, HVAC, and Warehouse departments at its injectable manufacturing facility in Navsari, Gujarat. The company is offering Officer and Senior Officer level roles for candidates with 2 to 8 years of experience in pharmaceutical manufacturing, particularly in sterile injectable and lyophilized products.
This is a strong opportunity for professionals from B.Pharm, M.Pharm, M.Sc, B.Sc, and engineering backgrounds who are looking to build long-term careers in GMP-compliant pharmaceutical environments, especially within injectable manufacturing, microbiology testing, and plant engineering systems.
Company Overview
Gufic Biosciences Ltd is a well-established pharmaceutical company with over five decades of industry presence, specializing in lyophilized injectables and critical care products. The company operates advanced manufacturing facilities with global regulatory approvals including WHO-GMP, EU-GMP, ANVISA, Canada, Russia, and other international accreditations.
Known for its expertise in sterile manufacturing, API development, and innovative drug delivery systems, Gufic Biosciences plays a significant role in improving healthcare outcomes. The Navsari facility is equipped with modern infrastructure for injectable production, quality testing, and regulatory compliance, making it a preferred workplace for pharma professionals.
Working with Gufic provides exposure to global regulatory frameworks, aseptic processing technologies, and high-quality pharmaceutical manufacturing practices.
Job Role & Responsibilities
Quality Assurance (Injectables – IPQA & Compliance)
- Perform in-process quality assurance (IPQA) activities in injectable manufacturing
- Handle QMS documentation including deviations, CAPA, change control, and audit compliance
- Conduct validation activities and vendor qualification processes
- Ensure compliance with GLP, cGMP, and regulatory guidelines
- Participate in internal and external audits and ensure documentation readiness
Quality Control (Microbiology)
- Perform microbiological testing including sterility, BET, MLT, and environmental monitoring
- Handle culture preparation, media preparation, and microbial analysis
- Maintain laboratory records and ensure compliance with microbiology standards
- Support validation and qualification of microbiological methods
Production (Injectables – Aseptic Operations)
- Handle aseptic manufacturing processes including lyophilization, SVP/LVP lines, PFS, and cartridge filling
- Maintain batch records and production documentation
- Ensure adherence to GMP and sterile manufacturing practices
- Support process optimization and troubleshooting in production
Maintenance (Engineering)
- Perform preventive and breakdown maintenance of plant equipment
- Handle utility systems including water systems, air compressors, chillers, and cooling towers
- Ensure smooth operation of manufacturing equipment and utilities
HVAC (Engineering Systems)
- Operate and maintain HVAC systems, BMS, PLC, and SCADA
- Monitor environmental conditions in manufacturing areas
- Ensure compliance with cleanroom standards and regulatory requirements
Warehouse (Materials Management)
- Manage raw material and packing material dispensing (RM/PM)
- Maintain inward registers, GRN, and SAP documentation
- Ensure compliance with GMP and regulatory requirements in warehouse operations
Eligibility / Qualifications
Candidates must have completed:
B.Pharm, M.Pharm, B.Sc, M.Sc, Diploma Mechanical, BE/ME Mechanical
Relevant courses include: Pharmaceutical Analysis, Microbiology, Biotechnology, Pharmaceutics, Industrial Pharmacy, Mechanical Engineering, Production Technology, Quality Assurance, Regulatory Affairs
- Experience: 2 to 8 years depending on department
- Strong knowledge of injectable manufacturing, aseptic practices, and GMP compliance
- Experience in regulated plants (WHO-GMP, EU-GMP, etc.) preferred
- Technical expertise in microbiology, analytical testing, or engineering systems based on role
- Knowledge of SAP and documentation systems is an added advantage
Location & Salary
- Job Location: Gufic Biosciences Ltd., Navsari, Gujarat
- Salary: Competitive and aligned with industry standards based on experience
Application Process
Interested candidates can apply by sending their updated resume to:
Email: binal.kapadia@guficbio.com
Contact Number: +91 63599 21281
Important Instructions:
- Apply within 10 working days
- Ensure your CV highlights relevant experience in injectable manufacturing, microbiology, QA/QC, or engineering
- Candidates with regulatory plant exposure will be preferred
Why This Opportunity Stands Out
- Work with a leading injectable pharmaceutical company with global approvals
- Exposure to lyophilization technology and sterile manufacturing processes
- Strong career growth in QA, QC, production, and engineering roles
- Hands-on experience in GMP, regulatory compliance, and validation systems
- Opportunity to contribute to critical healthcare products and patient safety
FAQs
Who can apply for these roles?
Candidates with B.Pharm, M.Pharm, M.Sc, B.Sc, or engineering qualifications can apply.
Is injectable experience required?
Yes, candidates with injectable or sterile manufacturing experience are preferred.
What roles are available?
Quality Assurance, Quality Control (Microbiology), Production, Maintenance, HVAC, and Warehouse.
What is the experience requirement?
2 to 8 years depending on the department.
How to apply?
Send your updated resume to the provided email ID.
Summary Table
| Category | Details |
|---|---|
| Company | Gufic Biosciences Ltd. |
| Vacancies | QA Officer, QC Microbiologist, Production Executive, Maintenance Engineer, HVAC Engineer, Warehouse Officer |
| Required Education | B.Pharm, M.Pharm, B.Sc, M.Sc, Diploma Mechanical, BE/ME Mechanical |
| Experience | 2 to 8 Years |
You must sign in to apply for this position.


