Zenotech Hiring Assistant Manager – Regulatory Affairs / Liaisoning

- Company Overview
- Job Role & Responsibilities
- Assistant Manager – Regulatory Affairs / Liaisoning
- Eligibility / Qualifications
- Required Education
- Experience Requirements
- Location & Salary
- Location:
- Salary:
- Application Process
- Why This Opportunity Stands Out
- FAQs
- What does a Regulatory Affairs Assistant Manager do?
- Is FDA experience required?
- What is the experience requirement?
- Is this a senior-level role?
- How to apply?
- Summary Table
Senior Regulatory Affairs Jobs in Hyderabad – Pharma Assistant Manager Role Open with 10+ Years Experience
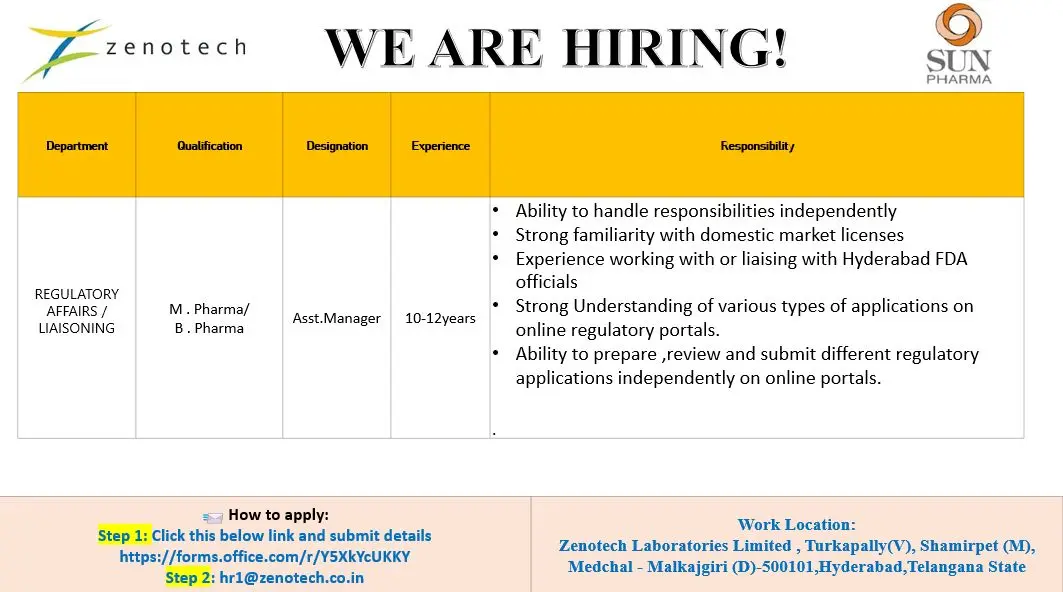
Zenotech Laboratories Limited has announced a senior-level hiring opportunity in the Regulatory Affairs / Liaisoning department for the position of Assistant Manager. This role is based in Hyderabad at the company’s manufacturing facility in Turkapally, Shamirpet. The company is specifically looking for experienced professionals with 10–12 years of regulatory experience, making it a high-value career move for candidates in pharma compliance and licensing.
This position is ideal for professionals who have hands-on experience in handling domestic regulatory approvals, working with FDA authorities, and managing online regulatory submissions. The role offers strong exposure to pharmaceutical regulatory systems, compliance frameworks, and government liaisoning activities.
Company Overview
Zenotech Laboratories Limited is a well-established pharmaceutical company specializing in oncology and biotechnology-based products. The company has built a strong reputation in regulated markets and continues to expand its presence in both domestic and international sectors.
With advanced manufacturing capabilities and a focus on compliance, Zenotech operates under strict regulatory frameworks. The company’s Hyderabad facility plays a key role in manufacturing and regulatory operations, making it an important hub for pharmaceutical professionals.
Working at Zenotech provides exposure to:
- Regulatory affairs and licensing processes
- Government liaisoning and compliance activities
- Online regulatory submission systems
- Pharmaceutical manufacturing and quality environments
This environment is ideal for experienced professionals aiming to grow in regulatory leadership roles.
Job Role & Responsibilities
Assistant Manager – Regulatory Affairs / Liaisoning
This role involves managing regulatory processes, handling licensing requirements, and coordinating with government authorities.
Key responsibilities include:
- Independently handle regulatory responsibilities and submissions
- Prepare, review, and submit applications on online regulatory portals
- Liaise with Hyderabad FDA officials for approvals and compliance
- Manage domestic market licenses and regulatory documentation
- Ensure adherence to regulatory guidelines and compliance standards
- Coordinate with internal teams for documentation and approvals
This role is critical in ensuring that pharmaceutical operations comply with legal and regulatory requirements.
Eligibility / Qualifications
Required Education
Candidates must have a pharmaceutical background.
Accepted courses include:
B.Pharm, M.Pharm, Pharmaceutical Sciences
Experience Requirements
- 10 to 12 years of experience in regulatory affairs or liaisoning
- Strong knowledge of domestic regulatory systems
- Experience working with FDA authorities and regulatory portals
- Ability to handle regulatory responsibilities independently
Location & Salary
Location:
- Zenotech Laboratories Limited, Turkapally (V), Shamirpet (M), Medchal-Malkajgiri District, Hyderabad, Telangana
Salary:
- Salary is not disclosed
- Senior regulatory roles typically offer high compensation with strong growth potential in pharma industry
Application Process
Interested candidates can apply through the following steps:
Step 1: Submit details via online form:
https://forms.office.com/r/Y5XkYcUKKY
Step 2: Send your resume to:
Candidates should ensure:
- Resume highlights regulatory experience and FDA liaisoning
- Mention experience with online regulatory portals
- Include details of licensing and compliance work
Why This Opportunity Stands Out
- Senior-level regulatory role with leadership responsibilities
- Direct interaction with regulatory authorities
- Exposure to licensing and compliance systems
- Strong career growth in regulatory affairs domain
This role is ideal for experienced professionals aiming to advance in regulatory affairs, pharma compliance, and government liaisoning roles.
FAQs
What does a Regulatory Affairs Assistant Manager do?
This role involves handling regulatory submissions, licensing, and compliance activities while coordinating with authorities.
Is FDA experience required?
Yes, experience working with regulatory authorities like FDA is important for this role.
What is the experience requirement?
Candidates should have 10–12 years of experience in regulatory affairs.
Is this a senior-level role?
Yes, this is a senior-level position with independent responsibilities.
How to apply?
Candidates must submit details via the online form and send their resume via email.
Summary Table
| Company | Zenotech Laboratories Limited |
|---|---|
| Vacancies | Assistant Manager – Regulatory Affairs / Liaisoning |
| Required Education | B.Pharm, M.Pharm, Pharmaceutical Sciences |
| Experience | 10–12 years in Regulatory Affairs |
To apply for this job please visit forms.office.com.


